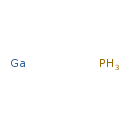Gallium phosphide (CHEM001999)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-07-02 16:02:14 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:39 UTC | |||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001999 | |||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Gallium phosphide | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||
| Description | Gallium phosphide is a phosphide of gallium. It is a semiconductor and optical material that is used for manufacture of low and standard brightness red, orange, and green light-emitting diodes. Metal phosphides are hydrolysed to phosphine upon contact with water or stomach acid. Phosphine is a colorless, flammable, explosive, and toxic gas. (3, 5) | |||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | GaH3P | |||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 103.721 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 102.923 g/mol | |||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 12063-98-8 | |||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | gallane phosphane | |||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | gallane phosphane | |||||||||||||||||||||||||||||||||||||||||||||
| SMILES | P.[Ga] | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/Ga.H3P/h;1H3 | |||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | ZETOOLHODJOIKC-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as miscellaneous mixed metal/non-metals. These are inorganic compounds containing non-metal as well as metal atoms but not belonging to afore mentioned classes. | |||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Mixed metal/non-metal compounds | |||||||||||||||||||||||||||||||||||||||||||||
| Class | Miscellaneous mixed metal/non-metals | |||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Miscellaneous mixed metal/non-metals | |||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Orange solid. | |||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (4) ; inhalation (4) | |||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Phosphine inhibits cytochrome c oxidase, preventing mitochondrial oxidative phosphorylation. This non-competitive inhibition prevents cellular respiration and leads to multi-organ dysfunction. Phosphine can also react with hydrogen peroxide to form the highly reactive hydroxyl radical, which can cause lipid peroxidation. (1, 2) | |||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Phosphine and metal phosphides may be absorbed following ingestion or inhalation, then distribute to the nervous system, liver, and kidney. In the body, metal phosphides are hydrolysed to phosphine, and phosphine is oxidized to hypophosphite and phosphite. Metabolites are excreted in the urine, while unchanged phosphine is exhaled. (4) | |||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | |||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Gallium phosphide is a semiconductor and optical material that is used for manufacture of low and standard brightness red, orange, and green light-emitting diodes. (5) | |||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Inhalation of phosphine may cause severe pulmonary irritation leading to acute pulmonary oedema, cardiovascular dysfunction, CNS excitation, coma and death. Gastrointestinal disorders, renal damage and leukopenia may also occur. Chronic exposure to phosphine can result in anemia, bronchitis, gastrointestinal effects, and visual, speech and motor problems. (3, 4) | |||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Early symptoms of acute phosphine intoxication include pain in the diaphragm, nausea, vomiting, excitement, and a phosphorus smell on the breath. Higher levels can cause weakness, bronchitis, pulmonary edema, shortness of breath, convulsions, and death. Some effects, such as pulmonary edema, convulsions, and liver injury, may appear or continue to be present days after an exposure. Ingestion of metal phosphides results in release of phosphine in your stomach which can cause nausea, vomiting, abdominal pain, and diarrhea. (3) | |||||||||||||||||||||||||||||||||||||||||||||
| Treatment | As there is no antidote for phosphine poisoning, treatment is mainly symptomatic. Artificial respiration, gastric lavage, and/or administration of activated charcoal may be necessary. (4) | |||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 19910168 | |||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||