| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 13:46:38 UTC |
|---|
| Update Date | 2016-11-09 01:23:25 UTC |
|---|
| Accession Number | CHEM045872 |
|---|
| Identification |
|---|
| Common Name | Sultopride |
|---|
| Class | Small Molecule |
|---|
| Description | Sultopride is used in Japan, Hong Kong, and Europe to treat schizophrenia. It is of the drug class atypical antipsychotics [L5626]. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
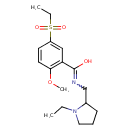
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Sultopride HCL | ChEMBL | | Barnetil | MeSH | | N-(Ethyl-1-pyrrolidinyl- 2-methyl)methoxy-2-ethylsulfonyl-5-benzamide | MeSH | | Solian | MeSH | | Amisulpride | MeSH | | Sultopride hydrochloride | MeSH | | 4-Amino-N-((1-ethyl-2-pyrrolidinyl)methyl)-5-(ethylsulfonyl)-2-methoxybenzamide | MeSH | | 5-(Ethanesulfonyl)-N-[(1-ethylpyrrolidin-2-yl)methyl]-2-methoxybenzene-1-carboximidate | Generator | | 5-(Ethanesulphonyl)-N-[(1-ethylpyrrolidin-2-yl)methyl]-2-methoxybenzene-1-carboximidate | Generator | | 5-(Ethanesulphonyl)-N-[(1-ethylpyrrolidin-2-yl)methyl]-2-methoxybenzene-1-carboximidic acid | Generator |
|
|---|
| Chemical Formula | C17H26N2O4S |
|---|
| Average Molecular Mass | 354.470 g/mol |
|---|
| Monoisotopic Mass | 354.161 g/mol |
|---|
| CAS Registry Number | 53583-79-2 |
|---|
| IUPAC Name | 5-(ethanesulfonyl)-N-[(1-ethylpyrrolidin-2-yl)methyl]-2-methoxybenzene-1-carboximidic acid |
|---|
| Traditional Name | sultopride |
|---|
| SMILES | CCN1CCCC1CN=C(O)C1=C(OC)C=CC(=C1)S(=O)(=O)CC |
|---|
| InChI Identifier | InChI=1S/C17H26N2O4S/c1-4-19-10-6-7-13(19)12-18-17(20)15-11-14(24(21,22)5-2)8-9-16(15)23-3/h8-9,11,13H,4-7,10,12H2,1-3H3,(H,18,20) |
|---|
| InChI Key | UNRHXEPDKXPRTM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzenesulfonyl compounds. These are aromatic compounds containing a benzenesulfonyl group, which consists of a monocyclic benzene moiety that carries a sulfonyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzenesulfonyl compounds |
|---|
| Direct Parent | Benzenesulfonyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzenesulfonyl group
- Phenoxy compound
- Anisole
- Phenol ether
- Methoxybenzene
- Alkyl aryl ether
- N-alkylpyrrolidine
- Pyrrolidine
- Sulfone
- Sulfonyl
- Tertiary amine
- Tertiary aliphatic amine
- Carboximidic acid
- Carboximidic acid derivative
- Ether
- Azacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organic nitrogen compound
- Amine
- Organic oxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-004j-9012000000-bedc5cc06b419b38804c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0bvi-1619000000-5ca12040b521e01f0d2c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03fr-2921000000-c12a9fc8a715ca2aa1b2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01qm-9300000000-6ddd6c00479109f87753 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0009000000-219cc8f6dc4e1d7c900f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0fbc-1449000000-7565e61fe91c31832a63 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9531000000-fa8a4a6720a1e5c05b33 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB13273 |
|---|
| HMDB ID | HMDB0258623 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Sultopride |
|---|
| Chemspider ID | 5164 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | C11708 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|