| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 13:33:49 UTC |
|---|
| Update Date | 2016-11-09 01:23:23 UTC |
|---|
| Accession Number | CHEM045660 |
|---|
| Identification |
|---|
| Common Name | azilsartan |
|---|
| Class | Small Molecule |
|---|
| Description | A benzimidazolecarboxylic acid that is benzimidazole-7-carboxylic acid substituted at position 2 by a methoxy group and at position 1 by a 2'-[(5-oxo-4,5-dihydro-1,2,4-oxadiazol-3-yl)biphenyl-4-yl]methyl group. Used (as the prodrug, azilsartan medoxomil) for treatment of hypertension. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
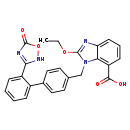
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| TAK 536 | ChEBI | | TAK-536 | ChEBI | | Azilva | Kegg | | 2-Ethoxy-1-((2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-biphenyl-4-yl)methyl)-1H-benzimidazole-7-carboxylic acid | MeSH | | 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-[1,1'-biphenyl]-4-yl]methyl}-1H-1,3-benzodiazole-7-carboxylate | Generator |
|
|---|
| Chemical Formula | C25H20N4O5 |
|---|
| Average Molecular Mass | 456.458 g/mol |
|---|
| Monoisotopic Mass | 456.143 g/mol |
|---|
| CAS Registry Number | 147403-03-0 |
|---|
| IUPAC Name | 2-ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-[1,1'-biphenyl]-4-yl]methyl}-1H-1,3-benzodiazole-7-carboxylic acid |
|---|
| Traditional Name | 2-ethoxy-3-{[2'-(5-oxo-2H-1,2,4-oxadiazol-3-yl)-[1,1'-biphenyl]-4-yl]methyl}-1,3-benzodiazole-4-carboxylic acid |
|---|
| SMILES | CCOC1=NC2=CC=CC(C(O)=O)=C2N1CC1=CC=C(C=C1)C1=CC=CC=C1C1=NC(=O)ON1 |
|---|
| InChI Identifier | InChI=1S/C25H20N4O5/c1-2-33-24-26-20-9-5-8-19(23(30)31)21(20)29(24)14-15-10-12-16(13-11-15)17-6-3-4-7-18(17)22-27-25(32)34-28-22/h3-13H,2,14H2,1H3,(H,30,31)(H,27,28,32) |
|---|
| InChI Key | KGSXMPPBFPAXLY-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as biphenyls and derivatives. These are organic compounds containing to benzene rings linked together by a C-C bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Biphenyls and derivatives |
|---|
| Direct Parent | Biphenyls and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Biphenyl
- Phenyl-1,2,4-oxadiazole
- Benzimidazole
- Alkyl aryl ether
- N-substituted imidazole
- 1,2,4-oxadiazole
- Azole
- Heteroaromatic compound
- Imidazole
- Oxadiazole
- Vinylogous amide
- Carboxylic acid derivative
- Carboxylic acid
- Ether
- Oxacycle
- Monocarboxylic acid or derivatives
- Azacycle
- Organoheterocyclic compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organonitrogen compound
- Organooxygen compound
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-0194500000-df27d34aa2f83656a214 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0010900000-32267dd1d4b63950bee2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03dr-0028900000-1765c453df741674e701 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-0096100000-9bf752eea1a45025244f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-08fr-0001900000-15e4bb7ab4f590efe40b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0102900000-c0617e7b705c6071beb2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0uxr-1139000000-d3fcd3cc57c3eff9efa3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4r-0000900000-93b0af2cfa3cb6391352 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-0013900000-1158bad8ebb3216b1001 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0zfr-0597100000-b41a92820f638c2c1d34 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0006900000-f08e2fd0336f593224dd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-02t9-0009400000-3df799b934a96ee951d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0gbc-2229200000-a30a382d4b50df751547 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Azilsartan |
|---|
| Chemspider ID | 8001032 |
|---|
| ChEBI ID | 68850 |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|