| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 12:45:10 UTC |
|---|
| Update Date | 2016-11-09 01:23:15 UTC |
|---|
| Accession Number | CHEM045003 |
|---|
| Identification |
|---|
| Common Name | Neo Heliopan® AP |
|---|
| Class | Small Molecule |
|---|
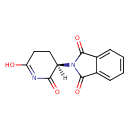
| Description | A 2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione that has R-configuration at the chiral centre. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (+)-Thalidomide | ChEBI | | (R)-(+)-Thalidomide | ChEBI | | D-Thalidomide | ChEBI | | R-(+)-Thalidomide | ChEBI |
|
|---|
| Chemical Formula | C13H10N2O4 |
|---|
| Average Molecular Mass | 258.233 g/mol |
|---|
| Monoisotopic Mass | 258.064 g/mol |
|---|
| CAS Registry Number | 2614-06-4 |
|---|
| IUPAC Name | 2-[(3R)-6-hydroxy-2-oxo-2,3,4,5-tetrahydropyridin-3-yl]-2,3-dihydro-1H-isoindole-1,3-dione |
|---|
| Traditional Name | 2-[(3R)-6-hydroxy-2-oxo-4,5-dihydro-3H-pyridin-3-yl]isoindole-1,3-dione |
|---|
| SMILES | [H][C@]1(CCC(O)=NC1=O)N1C(=O)C2=CC=CC=C2C1=O |
|---|
| InChI Identifier | InChI=1S/C13H10N2O4/c16-10-6-5-9(11(17)14-10)15-12(18)7-3-1-2-4-8(7)13(15)19/h1-4,9H,5-6H2,(H,14,16,17)/t9-/m1/s1 |
|---|
| InChI Key | UEJJHQNACJXSKW-SECBINFHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phthalimides. These are aromatic heterocyclic compounds containing a 1,3-dioxoisoindoline moiety. They are imide derivatives of phthalic anhydrides. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Isoindoles and derivatives |
|---|
| Sub Class | Isoindolines |
|---|
| Direct Parent | Phthalimides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phthalimide
- Alpha-amino acid or derivatives
- Isoindole
- Tetrahydropyridine
- Hydropyridine
- Carboxylic acid imide, n-substituted
- Benzenoid
- Carboxylic acid imide
- N-acylimine
- Lactim
- Azacycle
- Carboxylic acid derivative
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Organic nitrogen compound
- Organooxygen compound
- Organonitrogen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | - 2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione (CHEBI:61917 )
|
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0090000000-c87f93a626973b732cbf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4u-0490000000-b991e78be7b5b1bff22c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000l-9600000000-4dd354caec67b9f90b90 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0190000000-c2de70440450c7b134c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06rf-5590000000-44f28fd1a9fede3ad9a2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0007-9600000000-406d31db5a2e008a15d2 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 61917 |
|---|
| PubChem Compound ID | 75792 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|