| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 12:44:37 UTC |
|---|
| Update Date | 2016-11-09 01:23:15 UTC |
|---|
| Accession Number | CHEM044992 |
|---|
| Identification |
|---|
| Common Name | CEDREN-AC |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
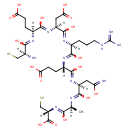
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (4S)-4-{[(2R)-2-amino-1-hydroxy-3-sulfanylpropylidene]amino}-4-{[(1S)-1-{[(1S)-4-carbamimidamido-1-{[(1S)-3-carboxy-1-{[(1S)-1-{[(1S)-1-{[(1R)-1-carboxy-2-sulfanylethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}-2-(C-hydroxycarbonimidoyl)ethyl]-C-hydroxycarbonimidoyl}propyl]-C-hydroxycarbonimidoyl}butyl]-C-hydroxycarbonimidoyl}-2-carboxyethyl]-C-hydroxycarbonimidoyl}butanoate | Generator | | (4S)-4-{[(2R)-2-amino-1-hydroxy-3-sulphanylpropylidene]amino}-4-{[(1S)-1-{[(1S)-4-carbamimidamido-1-{[(1S)-3-carboxy-1-{[(1S)-1-{[(1S)-1-{[(1R)-1-carboxy-2-sulphanylethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}-2-(C-hydroxycarbonimidoyl)ethyl]-C-hydroxycarbonimidoyl}propyl]-C-hydroxycarbonimidoyl}butyl]-C-hydroxycarbonimidoyl}-2-carboxyethyl]-C-hydroxycarbonimidoyl}butanoate | Generator | | (4S)-4-{[(2R)-2-amino-1-hydroxy-3-sulphanylpropylidene]amino}-4-{[(1S)-1-{[(1S)-4-carbamimidamido-1-{[(1S)-3-carboxy-1-{[(1S)-1-{[(1S)-1-{[(1R)-1-carboxy-2-sulphanylethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}-2-(C-hydroxycarbonimidoyl)ethyl]-C-hydroxycarbonimidoyl}propyl]-C-hydroxycarbonimidoyl}butyl]-C-hydroxycarbonimidoyl}-2-carboxyethyl]-C-hydroxycarbonimidoyl}butanoic acid | Generator |
|
|---|
| Chemical Formula | C33H54N12O16S2 |
|---|
| Average Molecular Mass | 938.980 g/mol |
|---|
| Monoisotopic Mass | 938.322 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (4S)-4-{[(2R)-2-amino-1-hydroxy-3-sulfanylpropylidene]amino}-4-{[(1S)-1-{[(1S)-4-carbamimidamido-1-{[(1S)-3-carboxy-1-{[(1S)-1-{[(1S)-1-{[(1R)-1-carboxy-2-sulfanylethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}-2-(C-hydroxycarbonimidoyl)ethyl]-C-hydroxycarbonimidoyl}propyl]-C-hydroxycarbonimidoyl}butyl]-C-hydroxycarbonimidoyl}-2-carboxyethyl]-C-hydroxycarbonimidoyl}butanoic acid |
|---|
| Traditional Name | (4S)-4-{[(2R)-2-amino-1-hydroxy-3-sulfanylpropylidene]amino}-4-{[(1S)-1-{[(1S)-4-carbamimidamido-1-{[(1S)-3-carboxy-1-{[(1S)-1-{[(1S)-1-{[(1R)-1-carboxy-2-sulfanylethyl]-C-hydroxycarbonimidoyl}ethyl]-C-hydroxycarbonimidoyl}-2-(C-hydroxycarbonimidoyl)ethyl]-C-hydroxycarbonimidoyl}propyl]-C-hydroxycarbonimidoyl}butyl]-C-hydroxycarbonimidoyl}-2-carboxyethyl]-C-hydroxycarbonimidoyl}butanoic acid |
|---|
| SMILES | [H][C@](N)(CS)C(O)=N[C@@]([H])(CCC(O)=O)C(O)=N[C@@]([H])(CC(O)=O)C(O)=N[C@@]([H])(CCCNC(N)=N)C(O)=N[C@@]([H])(CCC(O)=O)C(O)=N[C@@]([H])(CC(O)=N)C(O)=N[C@@]([H])(C)C(O)=N[C@@]([H])(CS)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C33H54N12O16S2/c1-13(25(53)45-20(12-63)32(60)61)39-30(58)18(9-21(35)46)43-29(57)17(5-7-23(49)50)42-27(55)15(3-2-8-38-33(36)37)41-31(59)19(10-24(51)52)44-28(56)16(4-6-22(47)48)40-26(54)14(34)11-62/h13-20,62-63H,2-12,34H2,1H3,(H2,35,46)(H,39,58)(H,40,54)(H,41,59)(H,42,55)(H,43,57)(H,44,56)(H,45,53)(H,47,48)(H,49,50)(H,51,52)(H,60,61)(H4,36,37,38)/t13-,14-,15-,16-,17-,18-,19-,20-/m0/s1 |
|---|
| InChI Key | VADZGSYPUYEZFI-QFHQRVFTSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as hybrid peptides. Hybrid peptides are compounds containing at least two different types of amino acids (alpha, beta, gamma, delta) linked to each other through a peptide bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Peptidomimetics |
|---|
| Sub Class | Hybrid peptides |
|---|
| Direct Parent | Hybrid peptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hybrid peptide
- Tetracarboxylic acid or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Cysteine or derivatives
- Alpha-amino acid or derivatives
- Amino acid
- Guanidine
- Amino acid or derivatives
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Carboximidamide
- Carboxylic acid
- Carboxylic acid derivative
- Carboximidic acid derivative
- Carboximidic acid
- Alkylthiol
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Imine
- Carbonyl group
- Amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0fmi-2301110139-1667e1d8b66330b4ee91 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00b9-9612200222-0d1d36c4272c859d38ce | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05i0-9632100010-0a0b46779f544548a927 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0ldr-1000000096-26b8fd9e0db0d1eb8ca4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0aor-3000000093-e04763d9fed99b0237c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9700113220-ae6ae2b675f557395842 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|