| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 12:31:07 UTC |
|---|
| Update Date | 2016-11-09 01:23:12 UTC |
|---|
| Accession Number | CHEM044726 |
|---|
| Identification |
|---|
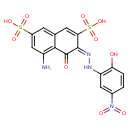
| Common Name | 5-amino-4-hydroxy-3-[(2-hydroxy-5-nitrophenyl)azo]naphthalene-2,7-disulfonic acid |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (3E)-5-Amino-3-[2-(2-hydroxy-5-nitrophenyl)hydrazin-1-ylidene]-4-oxo-3,4-dihydronaphthalene-2,7-disulfonate | Generator | | (3E)-5-Amino-3-[2-(2-hydroxy-5-nitrophenyl)hydrazin-1-ylidene]-4-oxo-3,4-dihydronaphthalene-2,7-disulphonate | Generator | | (3E)-5-Amino-3-[2-(2-hydroxy-5-nitrophenyl)hydrazin-1-ylidene]-4-oxo-3,4-dihydronaphthalene-2,7-disulphonic acid | Generator |
|
|---|
| Chemical Formula | C16H12N4O10S2 |
|---|
| Average Molecular Mass | 484.410 g/mol |
|---|
| Monoisotopic Mass | 483.999 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (3E)-5-amino-3-[2-(2-hydroxy-5-nitrophenyl)hydrazin-1-ylidene]-4-oxo-3,4-dihydronaphthalene-2,7-disulfonic acid |
|---|
| Traditional Name | (3E)-5-amino-3-[2-(2-hydroxy-5-nitrophenyl)hydrazin-1-ylidene]-4-oxonaphthalene-2,7-disulfonic acid |
|---|
| SMILES | NC1=C2C(=O)\C(=N/NC3=C(O)C=CC(=C3)N(=O)=O)C(=CC2=CC(=C1)S(O)(=O)=O)S(O)(=O)=O |
|---|
| InChI Identifier | InChI=1S/C16H12N4O10S2/c17-10-6-9(31(25,26)27)3-7-4-13(32(28,29)30)15(16(22)14(7)10)19-18-11-5-8(20(23)24)1-2-12(11)21/h1-6,18,21H,17H2,(H,25,26,27)(H,28,29,30)/b19-15- |
|---|
| InChI Key | UNJCYXUDAPTLRZ-CYVLTUHYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as naphthalenes. Naphthalenes are compounds containing a naphthalene moiety, which consists of two fused benzene rings. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Naphthalenes |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Naphthalenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Nitrophenol
- Naphthalene
- 1-sulfo,2-unsubstituted aromatic compound
- Nitrobenzene
- Arylsulfonic acid or derivatives
- Nitroaromatic compound
- Aryl ketone
- Phenylhydrazine
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Monocyclic benzene moiety
- Vinylogous amide
- Sulfonyl
- Organosulfonic acid
- Organosulfonic acid or derivatives
- Organic sulfonic acid or derivatives
- Organic nitro compound
- C-nitro compound
- Ketone
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Allyl-type 1,3-dipolar organic compound
- Organic oxoazanium
- Hydrazone
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Primary amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Amine
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014r-0000900000-be3ef86f8cd9ff244aca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0kmi-0396400000-2bc236c021400b168868 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-3942000000-679878056d6e415be851 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0111900000-e70568652a1fcf62218c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ugi-2492500000-ab918def5d76c5fae651 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01q9-5960000000-1e2c3d19fa0caef35add | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 9575979 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|