| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 12:09:54 UTC |
|---|
| Update Date | 2016-11-09 01:23:07 UTC |
|---|
| Accession Number | CHEM044392 |
|---|
| Identification |
|---|
| Common Name | 2,2'-[(2,2',5,5'-tetrachloro[1,1'-biphenyl]-4,4'-diyl)bis(azo)]bis[N-(2,4-dimethylphenyl)-3-oxobutyramide] |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
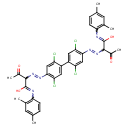
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| N-(2,4-Dimethylphenyl)-3-oxo-2-{2-[2,2',5,5'-tetrachloro-4'-(2-{1-[(2,4-dimethylphenyl)-C-hydroxycarbonimidoyl]-2-oxopropyl}diazen-1-yl)-[1,1'-biphenyl]-4-yl]diazen-1-yl}butanimidate | Generator |
|
|---|
| Chemical Formula | C36H32Cl4N6O4 |
|---|
| Average Molecular Mass | 754.490 g/mol |
|---|
| Monoisotopic Mass | 752.124 g/mol |
|---|
| CAS Registry Number | 22094-93-5 |
|---|
| IUPAC Name | N-(2,4-dimethylphenyl)-3-oxo-2-{2-[2,2',5,5'-tetrachloro-4'-(2-{1-[(2,4-dimethylphenyl)-C-hydroxycarbonimidoyl]-2-oxopropyl}diazen-1-yl)-[1,1'-biphenyl]-4-yl]diazen-1-yl}butanimidic acid |
|---|
| Traditional Name | N-(2,4-dimethylphenyl)-3-oxo-2-{2-[2,2',5,5'-tetrachloro-4'-(2-{1-[(2,4-dimethylphenyl)-C-hydroxycarbonimidoyl]-2-oxopropyl}diazen-1-yl)-[1,1'-biphenyl]-4-yl]diazen-1-yl}butanimidic acid |
|---|
| SMILES | CC(=O)C(N=NC1=C(Cl)C=C(C(Cl)=C1)C1=CC(Cl)=C(C=C1Cl)N=NC(C(C)=O)C(O)=NC1=C(C)C=C(C)C=C1)C(O)=NC1=C(C)C=C(C)C=C1 |
|---|
| InChI Identifier | InChI=1S/C36H32Cl4N6O4/c1-17-7-9-29(19(3)11-17)41-35(49)33(21(5)47)45-43-31-15-25(37)23(13-27(31)39)24-14-28(40)32(16-26(24)38)44-46-34(22(6)48)36(50)42-30-10-8-18(2)12-20(30)4/h7-16,33-34H,1-6H3,(H,41,49)(H,42,50) |
|---|
| InChI Key | GPWXTTPSHZOIKO-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as benzidines. These are organic compounds containing the benzidine skeleton, made up of a biphenyl ring system substituted at the 4- and 4'-positions with a unsubstituted amine group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Biphenyls and derivatives |
|---|
| Direct Parent | Benzidines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Polychlorinated biphenyl
- Benzidine
- Chlorinated biphenyl
- Alpha-amino acid amide
- Alpha-amino acid or derivatives
- Anilide
- 1,4-dichlorobenzene
- N-arylamide
- M-xylene
- Xylene
- Halobenzene
- Chlorobenzene
- Aryl halide
- 1,3-dicarbonyl compound
- Aryl chloride
- Fatty acyl
- Fatty amide
- Carboxamide group
- Secondary carboxylic acid amide
- Ketone
- Azo compound
- Organic 1,3-dipolar compound
- Carboxylic acid derivative
- Propargyl-type 1,3-dipolar organic compound
- Organooxygen compound
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organonitrogen compound
- Organochloride
- Organohalogen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_4) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_5) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_6) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_7) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_8) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0uk9-0820010900-69109360eb59079c8421 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0900000000-6ed22579c52e2e28177e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-3900000000-6f8ae432c8689efc5176 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0gc0-0191162500-205b783e5a0ec5a27e48 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0w90-0460395500-d52ec765b496b651c2c2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01b9-3932100000-4a5702eafa5f65c7a9da | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0260301 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 21172832 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|