| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 11:01:00 UTC |
|---|
| Update Date | 2016-11-09 01:22:56 UTC |
|---|
| Accession Number | CHEM043529 |
|---|
| Identification |
|---|
| Common Name | ajamalicine |
|---|
| Class | Small Molecule |
|---|
| Description | A monoterpenoid indole alkaloid with formula C21H24N2O3, isolated from several Rauvolfia and Catharanthus species. It is a selective alpha1-adrenoceptor antagonist used for the treatment of high blood pressure. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
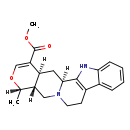
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (19alpha)-16,17-Didehydro-19-methyl-oxayohimban-16-carboxylic acid methyl ester | ChEBI | | Ajmalicin | ChEBI | | Circolene | ChEBI | | Cristanyl | ChEBI | | delta-Yohimbine | ChEBI | | Duxil | ChEBI | | Lamuran | ChEBI | | Methyl (4S,4ar,13BS,14as)-4-methyl-4a,5,7,8,13,13b,14,14a-octahydro-4H-indolo[2,3-a]pyrano[3,4-g]quinolizine-1-carboxylate | ChEBI | | Raubasine | ChEBI | | Sarpan | ChEBI | | (19a)-16,17-Didehydro-19-methyl-oxayohimban-16-carboxylate methyl ester | Generator | | (19a)-16,17-Didehydro-19-methyl-oxayohimban-16-carboxylic acid methyl ester | Generator | | (19alpha)-16,17-Didehydro-19-methyl-oxayohimban-16-carboxylate methyl ester | Generator | | (19Α)-16,17-didehydro-19-methyl-oxayohimban-16-carboxylate methyl ester | Generator | | (19Α)-16,17-didehydro-19-methyl-oxayohimban-16-carboxylic acid methyl ester | Generator | | Δ-yohimbine | Generator | | Methyl (4S,4ar,13BS,14as)-4-methyl-4a,5,7,8,13,13b,14,14a-octahydro-4H-indolo[2,3-a]pyrano[3,4-g]quinolizine-1-carboxylic acid | Generator | | Ajmalicine | KEGG | | Akuammigine | MeSH | | Ajmalicine, (19beta)-isomer | MeSH | | Rauvasan | MeSH | | Ajmalicine, (hydrochloride(19beta,20alpha))-isomer | MeSH | | tetrahydro-Alstonine | MeSH | | Ajmalicine, (3beta,19alpha,20alpha)-isomer | MeSH | | 19-Epiajmalicine | MeSH | | Tetrahydroalstonine | MeSH | | Ajmalicine, (19alpha,20alpha)-isomer | MeSH | | Ajmalicine, (19beta,20alpha)-isomer | MeSH | | Ajmalicine, (3-beta,19beta)-isomer | MeSH | | Ajmalicine, (3beta,19alpha)-isomer | MeSH | | Ajmalicine, PO4(19alpha)-isomer | MeSH | | Ajmalicine, hydrochloride(19alpha)-isomer | MeSH | | Methyl (1S,15R,16S,20S)-16-methyl-17-oxa-3,13-diazapentacyclo[11.8.0.0²,¹⁰.0⁴,⁹.0¹⁵,²⁰]henicosa-2(10),4,6,8,18-pentaene-19-carboxylic acid | Generator |
|
|---|
| Chemical Formula | C21H24N2O3 |
|---|
| Average Molecular Mass | 352.434 g/mol |
|---|
| Monoisotopic Mass | 352.179 g/mol |
|---|
| CAS Registry Number | 483-04-5 |
|---|
| IUPAC Name | methyl (1S,15R,16S,20S)-16-methyl-17-oxa-3,13-diazapentacyclo[11.8.0.0^{2,10}.0^{4,9}.0^{15,20}]henicosa-2(10),4,6,8,18-pentaene-19-carboxylate |
|---|
| Traditional Name | methyl (1S,15R,16S,20S)-16-methyl-17-oxa-3,13-diazapentacyclo[11.8.0.0^{2,10}.0^{4,9}.0^{15,20}]henicosa-2(10),4,6,8,18-pentaene-19-carboxylate |
|---|
| SMILES | [H][C@@]1(C)OC=C(C(=O)OC)[C@@]2([H])C[C@]3([H])N(CCC4=C3NC3=CC=CC=C43)C[C@]12[H] |
|---|
| InChI Identifier | InChI=1S/C21H24N2O3/c1-12-16-10-23-8-7-14-13-5-3-4-6-18(13)22-20(14)19(23)9-15(16)17(11-26-12)21(24)25-2/h3-6,11-12,15-16,19,22H,7-10H2,1-2H3/t12-,15-,16+,19-/m0/s1 |
|---|
| InChI Key | GRTOGORTSDXSFK-XJTZBENFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as yohimbine alkaloids. These are alkaloids containing the pentacyclic yohimban skeleton. The Yohimbinoid alkaloids contain a carbocyclic ring E arising through C-17 to C-18 bond formation in a corynantheine precursor. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Yohimbine alkaloids |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Yohimbine alkaloids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Ajmalicine-skeleton
- 18-oxayohimban
- Corynanthean skeleton
- Yohimbine alkaloid
- Beta-carboline
- Pyridoindole
- 3-alkylindole
- Indole
- Indole or derivatives
- Aralkylamine
- Benzenoid
- Piperidine
- Vinylogous ester
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Methyl ester
- Heteroaromatic compound
- Pyrrole
- Amino acid or derivatives
- Carboxylic acid ester
- Tertiary aliphatic amine
- Tertiary amine
- Carboxylic acid derivative
- Azacycle
- Oxacycle
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Hydrocarbon derivative
- Organonitrogen compound
- Amine
- Organic oxygen compound
- Organooxygen compound
- Organopnictogen compound
- Carbonyl group
- Organic nitrogen compound
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0029000000-d03a748d66c3c683b217 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0gb9-0093000000-c60306ce258f8b45a901 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udj-4691000000-c9830f1a8a3eb57f5167 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0019000000-d00ca9fa39f91f00a1cd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0uxu-0089000000-c1c9b8df63140dd4fbe7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-046r-3190000000-183e8742be4d35271413 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00001678 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Ajmalicine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 2524 |
|---|
| PubChem Compound ID | 441975 |
|---|
| Kegg Compound ID | C09024 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|