| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 10:54:01 UTC |
|---|
| Update Date | 2016-11-09 01:22:55 UTC |
|---|
| Accession Number | CHEM043432 |
|---|
| Identification |
|---|
| Common Name | metabolite M30 of Thiacloprid |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| {N-[(6-chloropyridin-3-yl)methyl]-N-(2-sulfoethyl)-(C-hydroxycarbonimidoyl)amino}methanimidate | Generator | | {N-[(6-chloropyridin-3-yl)methyl]-N-(2-sulphoethyl)-(C-hydroxycarbonimidoyl)amino}methanimidate | Generator | | {N-[(6-chloropyridin-3-yl)methyl]-N-(2-sulphoethyl)-(C-hydroxycarbonimidoyl)amino}methanimidic acid | Generator |
|
|---|
| Chemical Formula | C10H13ClN4O5S |
|---|
| Average Molecular Mass | 336.750 g/mol |
|---|
| Monoisotopic Mass | 336.030 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | {N-[(6-chloropyridin-3-yl)methyl]-N-(2-sulfoethyl)-(C-hydroxycarbonimidoyl)amino}methanimidic acid |
|---|
| Traditional Name | {N-[(6-chloropyridin-3-yl)methyl]-N-(2-sulfoethyl)-(C-hydroxycarbonimidoyl)amino}methanimidic acid |
|---|
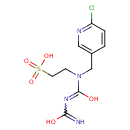
| SMILES | OC(=N)N=C(O)N(CCS(O)(=O)=O)CC1=CN=C(Cl)C=C1 |
|---|
| InChI Identifier | InChI=1S/C10H13ClN4O5S/c11-8-2-1-7(5-13-8)6-15(3-4-21(18,19)20)10(17)14-9(12)16/h1-2,5H,3-4,6H2,(H,18,19,20)(H3,12,14,16,17) |
|---|
| InChI Key | UCZRQNICFJGZAI-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2-halopyridines. These are organic compounds containing a pyridine ring substituted at the 2-position by a halogen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyridines and derivatives |
|---|
| Sub Class | Halopyridines |
|---|
| Direct Parent | 2-halopyridines |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2-halopyridine
- Aryl chloride
- Aryl halide
- Organic sulfonic acid or derivatives
- Organosulfonic acid or derivatives
- Heteroaromatic compound
- Organosulfonic acid
- Sulfonyl
- Alkanesulfonic acid
- Isourea
- Carboximidic acid derivative
- Azacycle
- Organic 1,3-dipolar compound
- Carboximidamide
- Propargyl-type 1,3-dipolar organic compound
- Organic oxide
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Organochloride
- Organohalogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Organic oxygen compound
- Imine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0269000000-5cff4ef455b475f35fbb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004l-0590000000-cb2e500225cef4de9992 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-1900000000-bf999b79c3834e073ca5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-9053000000-206f03e23749f108ddc8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0007-5090000000-aedd1ae0f80cae9bcfa7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01ox-9420000000-aa2bd6ceadd09e35b612 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|