| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 10:51:50 UTC |
|---|
| Update Date | 2016-11-09 01:22:54 UTC |
|---|
| Accession Number | CHEM043380 |
|---|
| Identification |
|---|
| Common Name | 2-Fluoroadenine 9-B-D-Arabinofuranoside |
|---|
| Class | Small Molecule |
|---|
| Description | Fludarabine is a chemotherapeutic agent used in the treatment of hematological malignancies. It is commonly marketed under the brand name Fludara. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
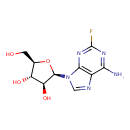
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Fluradosa | Kegg | | Fludarabine | MeSH | | F-Ara-a | MeSH | | 2-Fluoroadenine arabinoside | MeSH | | 9-beta-D-Arabinofuranosyl-2-fluoroadenine | MeSH |
|
|---|
| Chemical Formula | C10H12FN5O4 |
|---|
| Average Molecular Mass | 285.235 g/mol |
|---|
| Monoisotopic Mass | 285.087 g/mol |
|---|
| CAS Registry Number | 21679-14-1 |
|---|
| IUPAC Name | (2R,3S,4S,5R)-2-(6-amino-2-fluoro-9H-purin-9-yl)-5-(hydroxymethyl)oxolane-3,4-diol |
|---|
| Traditional Name | fludarabine |
|---|
| SMILES | NC1=NC(F)=NC2=C1N=CN2[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O |
|---|
| InChI Identifier | InChI=1S/C10H12FN5O4/c11-10-14-7(12)4-8(15-10)16(2-13-4)9-6(19)5(18)3(1-17)20-9/h2-3,5-6,9,17-19H,1H2,(H2,12,14,15)/t3-,5-,6+,9-/m1/s1 |
|---|
| InChI Key | HBUBKKRHXORPQB-FJFJXFQQSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as purine nucleosides. Purine nucleosides are compounds comprising a purine base attached to a ribosyl or deoxyribosyl moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Purine nucleosides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Purine nucleosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Purine nucleoside
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Pentose monosaccharide
- Imidazopyrimidine
- Purine
- Aminopyrimidine
- 2-halopyrimidine
- Halopyrimidine
- Aryl fluoride
- Aryl halide
- Monosaccharide
- N-substituted imidazole
- Imidolactam
- Pyrimidine
- Tetrahydrofuran
- Azole
- Heteroaromatic compound
- Imidazole
- Secondary alcohol
- Azacycle
- Organoheterocyclic compound
- Oxacycle
- Primary alcohol
- Amine
- Organic oxygen compound
- Organohalogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Organofluoride
- Alcohol
- Primary amine
- Organonitrogen compound
- Organic nitrogen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00r6-9670000000-e6d91df945dc377e1539 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-0059-0359000000-0759bfeee68a8abd65e2 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - LC-ESI-qTof , Positive | splash10-0udi-3900000000-5f778f763f3c6e93fd1e | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0udi-1920000000-6e5e3aa285e922e0f04a | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-0udi-3900000000-5f778f763f3c6e93fd1e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0940000000-b44d82f2edc7bb9b96e7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0900000000-fcdad1fd45b3f9ced41d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0zgi-1900000000-d768409f363b25a926b4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f80-0490000000-8fa587f3e22ff3c62c0d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0900000000-e2522f67fc384aab5bac | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f89-1900000000-e3268a920877eb1a4df4 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | DB01073 |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Fludarabine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 657237 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|