| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 10:16:32 UTC |
|---|
| Update Date | 2016-11-09 01:22:48 UTC |
|---|
| Accession Number | CHEM042807 |
|---|
| Identification |
|---|
| Common Name | nafoxidine |
|---|
| Class | Small Molecule |
|---|
| Description | Nafoxidine (INN; developmental code names U-11,000A, NSC-70735) or nafoxidine hydrochloride (USAN) is a nonsteroidal selective estrogen receptor modulator (SERM) or partial antiestrogen of the triphenylethylene group that was developed for the treatment of advanced breast cancer by Upjohn in the 1970s but was never marketed. It was developed at around the same time as tamoxifen and clomifene, which are also triphenylethylene derivatives. The drug was originally synthesized by the fertility control program at Upjohn as a postcoital contraceptive, but was subsequently repurposed for the treatment of breast cancer. Nafoxidine was assessed in clinical trials in the treatment of breast cancer and was found to be effective. However, it produced side effects including ichthyosis, partial hair loss, and phototoxicity of the skin in almost all patients, and this resulted in the discontinuation of its development.Nafoxidine is a long-acting estrogen receptor ligand, with a nuclear retention in the range of 24 to 48 hours or more. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
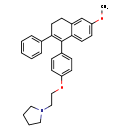
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Hydrochloride, nafoxidine | MeSH | | Nafoxidine hydrochloride | MeSH | | U-11000a | ChEMBL |
|
|---|
| Chemical Formula | C29H31NO2 |
|---|
| Average Molecular Mass | 425.572 g/mol |
|---|
| Monoisotopic Mass | 425.235 g/mol |
|---|
| CAS Registry Number | 1845-11-0 |
|---|
| IUPAC Name | 1-{2-[4-(6-methoxy-2-phenyl-3,4-dihydronaphthalen-1-yl)phenoxy]ethyl}pyrrolidine |
|---|
| Traditional Name | nafoxidine |
|---|
| SMILES | COC1=CC=C2C(CCC(C3=CC=CC=C3)=C2C2=CC=C(OCCN3CCCC3)C=C2)=C1 |
|---|
| InChI Identifier | InChI=1S/C29H31NO2/c1-31-26-14-16-28-24(21-26)11-15-27(22-7-3-2-4-8-22)29(28)23-9-12-25(13-10-23)32-20-19-30-17-5-6-18-30/h2-4,7-10,12-14,16,21H,5-6,11,15,17-20H2,1H3 |
|---|
| InChI Key | JEYWNNAZDLFBFF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylnaphthalenes. Phenylnaphthalenes are compounds containing a phenylnaphthalene skeleton, which consists of a naphthalene bound to a phenyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Naphthalenes |
|---|
| Sub Class | Phenylnaphthalenes |
|---|
| Direct Parent | Phenylnaphthalenes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylnaphthalene
- Stilbene
- Phenoxy compound
- Anisole
- Phenol ether
- Alkyl aryl ether
- Monocyclic benzene moiety
- N-alkylpyrrolidine
- Pyrrolidine
- Tertiary aliphatic amine
- Tertiary amine
- Ether
- Azacycle
- Organoheterocyclic compound
- Organooxygen compound
- Organonitrogen compound
- Organic nitrogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Amine
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-003r-9005100000-bf42a00371c7c92f6d61 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-2103900000-20d62335a037a2cecbb4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-9323100000-41f3b5ce93ea16447ea1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-9212000000-56de5f6b718fb9c79afd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0002900000-f87c247d1b57adee23b0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00fr-3029600000-2a34647b9a72227ebc9b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0201-5069000000-d8cb7647eeef6cefb87a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Nafoxidine |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 4416 |
|---|
| Kegg Compound ID | C14212 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|