| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-06-03 10:06:31 UTC |
|---|
| Update Date | 2016-11-09 01:22:47 UTC |
|---|
| Accession Number | CHEM042700 |
|---|
| Identification |
|---|
| Common Name | Bensulfuron methyl (free acid; IN-R9419) |
|---|
| Class | Small Molecule |
|---|
| Description | An N-sulfonylurea that is urea in which a hydrogen attached to one of the nitrogens has been replaced by an (o-carboxybenzyl)sulfonyl group, while a hydrogen attached to the other nitrogen has been replaced by a 4,6-dimethoxypyrimidin-2-yl group. An acetolactate synthase inhibitor, it is used (particularly as its methyl ester, bensulfuron-methyl) as a herbicide for the control of a variety of both annual and perennial weeds in crops, particularly wheat and rice. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
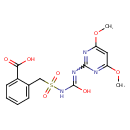
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-[[[[[(4,6-Dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]methyl]benzoic acid | ChEBI | | alpha-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluic acid | ChEBI | | 2-[[[[[(4,6-Dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulfonyl]methyl]benzoate | Generator | | 2-[[[[[(4,6-Dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulphonyl]methyl]benzoate | Generator | | 2-[[[[[(4,6-Dimethoxy-2-pyrimidinyl)amino]carbonyl]amino]sulphonyl]methyl]benzoic acid | Generator | | a-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluate | Generator | | a-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluic acid | Generator | | a-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluate | Generator | | a-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluic acid | Generator | | alpha-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluate | Generator | | alpha-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluate | Generator | | alpha-[(4,6-Dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluic acid | Generator | | Α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluate | Generator | | Α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulfamoyl]-O-toluic acid | Generator | | Α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluate | Generator | | Α-[(4,6-dimethoxypyrimidin-2-ylcarbamoyl)sulphamoyl]-O-toluic acid | Generator | | Bensulphuron | Generator |
|
|---|
| Chemical Formula | C15H16N4O7S |
|---|
| Average Molecular Mass | 396.370 g/mol |
|---|
| Monoisotopic Mass | 396.074 g/mol |
|---|
| CAS Registry Number | 99283-01-9 |
|---|
| IUPAC Name | 2-[({[(4,6-dimethoxypyrimidin-2-yl)-C-hydroxycarbonimidoyl]amino}sulfonyl)methyl]benzoic acid |
|---|
| Traditional Name | 2-({[(4,6-dimethoxypyrimidin-2-yl)-C-hydroxycarbonimidoyl]aminosulfonyl}methyl)benzoic acid |
|---|
| SMILES | COC1=CC(OC)=NC(N=C(O)NS(=O)(=O)CC2=CC=CC=C2C(O)=O)=N1 |
|---|
| InChI Identifier | InChI=1S/C15H16N4O7S/c1-25-11-7-12(26-2)17-14(16-11)18-15(22)19-27(23,24)8-9-5-3-4-6-10(9)13(20)21/h3-7H,8H2,1-2H3,(H,20,21)(H2,16,17,18,19,22) |
|---|
| InChI Key | PPWBRCCBKOWDNB-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pyrimidinyl-2-sulfonylureas. These are aromatic heterocyclic compounds containing a pyrimidine ring which is substituted with a sulfonylurea at the ring 2-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic nitrogen compounds |
|---|
| Class | Organonitrogen compounds |
|---|
| Sub Class | Sulfonylureas |
|---|
| Direct Parent | Pyrimidinyl-2-sulfonylureas |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pyrimidinyl-2-sulfonylurea
- Benzoic acid or derivatives
- Benzoic acid
- Benzoyl
- Alkyl aryl ether
- Monocyclic benzene moiety
- Benzenoid
- Pyrimidine
- Heteroaromatic compound
- Aminosulfonyl compound
- Sulfonyl
- Organosulfonic acid or derivatives
- Organic sulfonic acid or derivatives
- Carbonic acid derivative
- Azacycle
- Carboxylic acid derivative
- Carboxylic acid
- Ether
- Monocarboxylic acid or derivatives
- Organoheterocyclic compound
- Carbonyl group
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organic oxygen compound
- Organooxygen compound
- Organosulfur compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0f92-0739000000-718b0aabc4fc899a742d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0900000000-a234e6ea4d2e69c0f1f6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ukl-4900000000-df6cb47567a0956a2c4a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udj-0649000000-6edc58d9bfde61c4505f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-08mi-8892000000-74e73da809186cb3ab38 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-08i0-9720000000-570662387c78f493732a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 132880 |
|---|
| PubChem Compound ID | 107828 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|