| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 02:04:16 UTC |
|---|
| Update Date | 2016-11-09 01:22:41 UTC |
|---|
| Accession Number | CHEM042484 |
|---|
| Identification |
|---|
| Common Name | Catechol 1-O-sulphate |
|---|
| Class | Small Molecule |
|---|
| Description | An aryl sulfate that is catechol with one of the two hydroxy groups substituted by a sulfo group. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
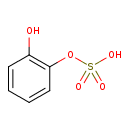
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2-Hydroxyphenyl)oxidanesulfonic acid | ChEBI | | 1,2-Benzenediol mono(hydrogen sulfate) | ChEBI | | 1,2-Benzenediol mono(hydrogen sulphate) | ChEBI | | Catechol monosulfate | ChEBI | | Catechol sulfate | ChEBI | | Pyrocatechol hydrogen sulfate | ChEBI | | Pyrocatechol monosulfate | ChEBI | | (2-Hydroxyphenyl)oxidanesulfonate | Generator | | (2-Hydroxyphenyl)oxidanesulphonate | Generator | | (2-Hydroxyphenyl)oxidanesulphonic acid | Generator | | 1,2-Benzenediol mono(hydrogen sulfuric acid) | Generator | | 1,2-Benzenediol mono(hydrogen sulphuric acid) | Generator | | Catechol monosulfuric acid | Generator | | Catechol monosulphate | Generator | | Catechol monosulphuric acid | Generator | | Catechol sulfuric acid | Generator | | Catechol sulphate | Generator | | Catechol sulphuric acid | Generator | | Pyrocatechol hydrogen sulfuric acid | Generator | | Pyrocatechol hydrogen sulphate | Generator | | Pyrocatechol hydrogen sulphuric acid | Generator | | Pyrocatechol monosulfuric acid | Generator | | Pyrocatechol monosulphate | Generator | | Pyrocatechol monosulphuric acid | Generator | | Pyrocatechol sulfuric acid | Generator | | Pyrocatechol sulphate | Generator | | Pyrocatechol sulphuric acid | Generator | | Catechol 1-O-sulfate | HMDB | | Catechol 1-O-sulfuric acid | HMDB | | Catechol 1-O-sulphuric acid | HMDB | | 2-Aminophenol sulfate | HMDB | | Pyrocatechol sulfate | HMDB |
|
|---|
| Chemical Formula | C6H6O5S |
|---|
| Average Molecular Mass | 190.174 g/mol |
|---|
| Monoisotopic Mass | 189.994 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2-hydroxyphenyl)oxidanesulfonic acid |
|---|
| Traditional Name | (2-hydroxyphenyl)oxidanesulfonic acid |
|---|
| SMILES | OC1=CC=CC=C1OS(O)(=O)=O |
|---|
| InChI Identifier | InChI=1S/C6H6O5S/c7-5-3-1-2-4-6(5)11-12(8,9)10/h1-4,7H,(H,8,9,10) |
|---|
| InChI Key | MZPWKJZDOCIALD-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylsulfates. Phenylsulfates are compounds containing a sulfuric acid group conjugated to a phenyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Organic sulfuric acids and derivatives |
|---|
| Sub Class | Arylsulfates |
|---|
| Direct Parent | Phenylsulfates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylsulfate
- Phenoxy compound
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Benzenoid
- Sulfuric acid ester
- Sulfate-ester
- Sulfuric acid monoester
- Monocyclic benzene moiety
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0bti-3900000000-e6f53d18ac22c7f5bfe9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00dr-6910000000-80237dcbd0d19fe9a392 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0900000000-e1185fdf71a2405723c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03dl-1900000000-f40e0d29b8b6a74dd3f4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-9100000000-dc63c933049bc6dd6775 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0900000000-1f7ed2ebb353e20269a0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-1900000000-d6dda28db9ba8ee670d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9800000000-36d37bd38c5d133fdcbc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0900000000-65e842416b97970070f9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-1900000000-a74fdd722d14d05c3f52 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-9300000000-2f6a02e142d2ef82baaa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01ox-0900000000-1590fbbb0009f5db5981 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01q9-9800000000-a3950de0d98e729d8c8f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0w29-9000000000-a4a9c033315ddd0f0d2d | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0059724 |
|---|
| FooDB ID | FDB031315 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 68505 |
|---|
| PubChem Compound ID | 3083879 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. https://www.ncbi.nlm.nih.gov/pubmed/?term=22770225 | | 2. Roux A, Xu Y, Heilier JF, Olivier MF, Ezan E, Tabet JC, Junot C: Annotation of the human adult urinary metabolome and metabolite identification using ultra high performance liquid chromatography coupled to a linear quadrupole ion trap-Orbitrap mass spectrometer. Anal Chem. 2012 Aug 7;84(15):6429-37. doi: 10.1021/ac300829f. Epub 2012 Jul 17. | | 3. Zhu Y, Wang P, Sha W, Sang S: Urinary Biomarkers of Whole Grain Wheat Intake Identified by Non-targeted and Targeted Metabolomics Approaches. Sci Rep. 2016 Nov 2;6:36278. doi: 10.1038/srep36278. | | 4. Publications of the University of Eastern Finland. Dissertations in Health Sciences., no 510 |
|
|---|