| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 02:04:15 UTC |
|---|
| Update Date | 2016-11-09 01:22:41 UTC |
|---|
| Accession Number | CHEM042483 |
|---|
| Identification |
|---|
| Common Name | 4-O-Methylgallic acid 3-O-sulphate |
|---|
| Class | Small Molecule |
|---|
| Description | 4-o-methylgallic acid 3-o-sulphate belongs to gallic acid and derivatives class of compounds. Those are compounds containing a 3,4,5-trihydroxybenzoic acid moiety. 4-o-methylgallic acid 3-o-sulphate is slightly soluble (in water) and an extremely strong acidic compound (based on its pKa). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
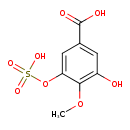
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4-Methylgallate 3-sulphate | Generator | | 4-Methylgallic acid 3-sulfuric acid | Generator | | 4-Methylgallic acid 3-sulphuric acid | Generator | | 4-O-Methylgallate 3-O-sulfate | HMDB | | 4-O-Methylgallate 3-O-sulphate | HMDB | | 4-O-Methylgallic acid 3-O-sulfuric acid | HMDB | | 4-O-Methylgallic acid 3-O-sulphuric acid | HMDB | | 4-O-Methylgallic acid-3-sulfate | HMDB | | 3-Hydroxy-4-methoxy-5-(sulfooxy)benzoic acid | HMDB | | 3-Hydroxy-4-methoxybenzoic acid-5-sulfate | HMDB |
|
|---|
| Chemical Formula | C8H8O8S |
|---|
| Average Molecular Mass | 264.200 g/mol |
|---|
| Monoisotopic Mass | 263.994 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 3-hydroxy-4-methoxy-5-(sulfooxy)benzoic acid |
|---|
| Traditional Name | 3-hydroxy-4-methoxy-5-(sulfooxy)benzoic acid |
|---|
| SMILES | COC1=C(OS(O)(=O)=O)C=C(C=C1O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C8H8O8S/c1-15-7-5(9)2-4(8(10)11)3-6(7)16-17(12,13)14/h2-3,9H,1H3,(H,10,11)(H,12,13,14) |
|---|
| InChI Key | LMJIEJLSDJBABY-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as gallic acid and derivatives. Gallic acid and derivatives are compounds containing a 3,4,5-trihydroxybenzoic acid moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | Gallic acid and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Gallic acid or derivatives
- P-methoxybenzoic acid or derivatives
- Phenylsulfate
- Methoxyphenol
- Arylsulfate
- Benzoic acid
- Phenoxy compound
- Methoxybenzene
- Phenol ether
- Benzoyl
- Anisole
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- 1-hydroxy-4-unsubstituted benzenoid
- Alkyl aryl ether
- Sulfuric acid ester
- Sulfate-ester
- Sulfuric acid monoester
- Organic sulfuric acid or derivatives
- Carboxylic acid derivative
- Carboxylic acid
- Ether
- Monocarboxylic acid or derivatives
- Organooxygen compound
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014j-0090000000-1a6b92ce7c09e4943fe2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014j-0890000000-d2c845dee70d469fac43 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01b9-9630000000-fcc4a22fcd87cf5f0dda | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-d5b7f2e1064926b0439f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-02u0-0980000000-c34a21284c7fb518622b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-015i-2900000000-7753b385097d204487c4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-e6c5943c9a289e37876b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03dj-3090000000-c88374065df74220def8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-9310000000-a224477b6019dd6f5e66 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0190000000-4da712a3901c50960b39 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-015i-0900000000-61d949f4818f28c109eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014r-6900000000-1d9993b5ed1e6fd526fa | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0240463 |
|---|
| FooDB ID | FDB031314 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 9000783 |
|---|
| ChEBI ID | 176487 |
|---|
| PubChem Compound ID | 10825483 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|