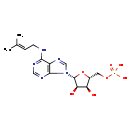
| Isopentenyl-AMP | ChEBI |
| Isopentenyladenosine riboside-5'-monophosphate | ChEBI |
| Isopentenyladenosine-5'-monophosphate | ChEBI |
| N-(3-Methyl-2-butenyl)-5'-adenylic acid | ChEBI |
| N6-(delta(2)-Isopentenyl)adenosine 5'-monophosphate | ChEBI |
| N6-(delta2-Isopentenyl)-adenosine 5'-monophosphate | ChEBI |
| N(6)-(Delta(2)-Isopentenyl)adenosine 5'-monophosphate | ChEBI |
| N(6)-(Dimethylallyl)adenosine 5'-(dihydrogen phosphate) | ChEBI |
| N6-(Dimethylallyl)adenosine 5'-phosphate | ChEBI |
| N(6)-(Dimethylallyl)adenosine 5'-phosphate | ChEBI |
| N(6)-Dimethylallyl-5'-adenylic acid | ChEBI |
| N(6)-Dimethylallyl-AMP | ChEBI |
| N(6)-Isopentenyladenosine-5'-monophosphate | ChEBI |
| Isopentenyladenosine riboside-5'-monophosphoric acid | Generator |
| Isopentenyladenosine-5'-monophosphoric acid | Generator |
| N-(3-Methyl-2-butenyl)-5'-adenylate | Generator |
| N6-(delta(2)-Isopentenyl)adenosine 5'-monophosphoric acid | Generator |
| N6-(Δ(2)-isopentenyl)adenosine 5'-monophosphate | Generator |
| N6-(Δ(2)-isopentenyl)adenosine 5'-monophosphoric acid | Generator |
| N6-(delta2-Isopentenyl)-adenosine 5'-monophosphoric acid | Generator |
| N6-(Δ2-isopentenyl)-adenosine 5'-monophosphate | Generator |
| N6-(Δ2-isopentenyl)-adenosine 5'-monophosphoric acid | Generator |
| N(6)-(delta(2)-Isopentenyl)adenosine 5'-monophosphoric acid | Generator |
| N(6)-(Δ(2)-isopentenyl)adenosine 5'-monophosphate | Generator |
| N(6)-(Δ(2)-isopentenyl)adenosine 5'-monophosphoric acid | Generator |
| N(6)-(Dimethylallyl)adenosine 5'-(dihydrogen phosphoric acid) | Generator |
| N6-(Dimethylallyl)adenosine 5'-phosphoric acid | Generator |
| N(6)-(Dimethylallyl)adenosine 5'-phosphoric acid | Generator |
| N(6)-Dimethylallyl-5'-adenylate | Generator |
| N(6)-Isopentenyladenosine-5'-monophosphoric acid | Generator |
| N(6)-(Dimethylallyl)adenosine 5'-monophosphoric acid | Generator |