| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:55:12 UTC |
|---|
| Update Date | 2016-11-09 01:22:37 UTC |
|---|
| Accession Number | CHEM042225 |
|---|
| Identification |
|---|
| Common Name | ferribactin |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
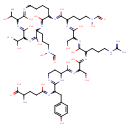
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Amino-4-{[1-(6-{[1-({4-carbamimidamido-1-[(2-hydroxy-1-{[4-(N-hydroxyformamido)-1-{[2,5,8,11-tetrahydroxy-3,6-bis(1-hydroxyethyl)-9-[3-(N-hydroxyformamido)propyl]-1,4,7,10-tetraazacyclohexadeca-1,4,7,10-tetraen-12-yl]-C-hydroxycarbonimidoyl}butyl]-C-hydroxycarbonimidoyl}ethyl)-C-hydroxycarbonimidoyl]butyl}-C-hydroxycarbonimidoyl)-2-hydroxyethyl]-C-hydroxycarbonimidoyl}-1,4,5,6-tetrahydropyrimidin-2-yl)-2-(4-hydroxyphenyl)ethyl]-C-hydroxycarbonimidoyl}butanoate | Generator |
|
|---|
| Chemical Formula | C56H90N18O21 |
|---|
| Average Molecular Mass | 1351.441 g/mol |
|---|
| Monoisotopic Mass | 1350.653 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 2-amino-4-[(1-{4-[(1-{[1-({1-[(1-{[3,6-bis(1-hydroxyethyl)-9-[3-(N-hydroxyformamido)propyl]-2,5,8,11-tetraoxo-1,4,7,10-tetraazacyclohexadecan-12-yl]carbamoyl}-4-(N-hydroxyformamido)butyl)carbamoyl]-2-hydroxyethyl}carbamoyl)-4-carbamimidamidobutyl]carbamoyl}-2-hydroxyethyl)carbamoyl]-1,4,5,6-tetrahydropyrimidin-2-yl}-2-(4-hydroxyphenyl)ethyl)carbamoyl]butanoic acid |
|---|
| Traditional Name | 2-amino-4-[(1-{4-[(1-{[1-({1-[(1-{[3,6-bis(1-hydroxyethyl)-9-[3-(N-hydroxyformamido)propyl]-2,5,8,11-tetraoxo-1,4,7,10-tetraazacyclohexadecan-12-yl]carbamoyl}-4-(N-hydroxyformamido)butyl)carbamoyl]-2-hydroxyethyl}carbamoyl)-4-carbamimidamidobutyl]carbamoyl}-2-hydroxyethyl)carbamoyl]-1,4,5,6-tetrahydropyrimidin-2-yl}-2-(4-hydroxyphenyl)ethyl)carbamoyl]butanoic acid |
|---|
| SMILES | CC(O)C1NC(=O)C(NC(=O)C(CCCN(O)C=O)NC(=O)C(CCCCNC1=O)NC(=O)C(CCCN(O)C=O)NC(=O)C(CO)NC(=O)C(CCCNC(N)=N)NC(=O)C(CO)NC(=O)C1CCNC(=N1)C(CC1=CC=C(O)C=C1)NC(=O)CCC(N)C(O)=O)C(C)O |
|---|
| InChI Identifier | InChI=1S/C56H90N18O21/c1-29(79)43-53(90)61-19-4-3-8-34(46(83)66-37(11-7-23-74(95)28-78)50(87)71-44(30(2)80)54(91)72-43)65-47(84)36(10-6-22-73(94)27-77)68-52(89)40(25-75)69-48(85)35(9-5-20-62-56(58)59)67-51(88)41(26-76)70-49(86)38-18-21-60-45(64-38)39(24-31-12-14-32(81)15-13-31)63-42(82)17-16-33(57)55(92)93/h12-15,27-30,33-41,43-44,75-76,79-81,94-95H,3-11,16-26,57H2,1-2H3,(H,60,64)(H,61,90)(H,63,82)(H,65,84)(H,66,83)(H,67,88)(H,68,89)(H,69,85)(H,70,86)(H,71,87)(H,72,91)(H,92,93)(H4,58,59,62) |
|---|
| InChI Key | XXKMKYGWXRCAHF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as cyclic peptides. Cyclic peptides are compounds containing a cyclic moiety bearing a peptide backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Cyclic peptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cyclic alpha peptide
- Alpha-amino acid
- Alpha-amino acid or derivatives
- Amphetamine or derivatives
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Benzenoid
- 1,4,5,6-tetrahydropyrimidine
- Hydropyrimidine
- Imidolactam
- Monocyclic benzene moiety
- Cyclic carboximidic acid
- Hydroxamic acid
- Amino acid or derivatives
- Guanidine
- Amino acid
- Secondary alcohol
- Amidine
- Carboxylic acid amidine
- Carboximidic acid
- Carboximidic acid derivative
- Carboxylic acid
- Azacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Carboximidamide
- Monocarboxylic acid or derivatives
- Polyol
- Organopnictogen compound
- Amine
- Alcohol
- Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Imine
- Primary aliphatic amine
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Organic oxygen compound
- Primary alcohol
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0029000000-094c4cd1887e93c852bc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0cdr-3179202111-6b490547e2168b50c71c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-092a-9678212041-956b30f590c16640da1d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udr-1049000000-6b4a7313bc401ddf2bad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udr-2089000000-580fc50e4dc0fa5215fa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0k96-9056204242-466913ee30018ab26bd7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0pb9-0029000000-c4f412ceeb9f9aee1800 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0029000100-a6b297279c4cfcb98b6b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6x-4291100010-b4a74798d08918a321c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0019000000-3d6f16938204ea1b6334 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0049000000-6e6a20e3029bb9865922 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9252010000-c2e696ed5e8fe371228f | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0304352 |
|---|
| FooDB ID | FDB030857 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|