| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:35:10 UTC |
|---|
| Update Date | 2016-11-09 01:22:30 UTC |
|---|
| Accession Number | CHEM041601 |
|---|
| Identification |
|---|
| Common Name | Equol 4'-O-glucuronide |
|---|
| Class | Small Molecule |
|---|
| Description | Equol 4'-O-glucuronide is a polyphenol metabolite detected in biological fluids (PMID: 20428313). |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
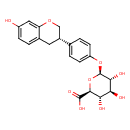
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2S,3S,4S,5R,6S)-3,4,5-Trihydroxy-6-{4-[(3S)-7-hydroxy-3,4-dihydro-2H-1-benzopyran-3-yl]phenoxy}oxane-2-carboxylate | HMDB |
|
|---|
| Chemical Formula | C21H22O9 |
|---|
| Average Molecular Mass | 418.394 g/mol |
|---|
| Monoisotopic Mass | 418.126 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2S,3S,4S,5R,6S)-3,4,5-trihydroxy-6-{4-[(3S)-7-hydroxy-3,4-dihydro-2H-1-benzopyran-3-yl]phenoxy}oxane-2-carboxylic acid |
|---|
| Traditional Name | (2S,3S,4S,5R,6S)-3,4,5-trihydroxy-6-{4-[(3S)-7-hydroxy-3,4-dihydro-2H-1-benzopyran-3-yl]phenoxy}oxane-2-carboxylic acid |
|---|
| SMILES | O[C@@H]1[C@@H](O)[C@H](OC2=CC=C(C=C2)[C@H]2COC3=CC(O)=CC=C3C2)O[C@@H]([C@H]1O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C21H22O9/c22-13-4-1-11-7-12(9-28-15(11)8-13)10-2-5-14(6-3-10)29-21-18(25)16(23)17(24)19(30-21)20(26)27/h1-6,8,12,16-19,21-25H,7,9H2,(H,26,27)/t12-,16+,17+,18-,19+,21-/m1/s1 |
|---|
| InChI Key | VIKIIYUHWNDRNI-LVEHSUOCSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as catechin gallates. These are organic compounds containing a gallate moiety glycosidically linked to a catechin. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavans |
|---|
| Direct Parent | Catechin gallates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Catechin gallate
- Flavonoid o-glycoside
- Epigallocatechin
- Flavonoid-7-o-glycoside
- 5-hydroxyflavonoid
- 3'-hydroxyflavonoid
- 4'-hydroxyflavonoid
- Hydroxyflavonoid
- Phenolic glycoside
- O-glucuronide
- 1-o-glucuronide
- Glucuronic acid or derivatives
- Gallic acid or derivatives
- M-hydroxybenzoic acid ester
- Glycosyl compound
- O-glycosyl compound
- Benzopyran
- Chromane
- Benzoate ester
- 1-benzopyran
- Benzenetriol
- Pyrogallol derivative
- Benzoic acid or derivatives
- Phenol ether
- Resorcinol
- Benzoyl
- Phenoxy compound
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Beta-hydroxy acid
- Phenol
- Monosaccharide
- Hydroxy acid
- Oxane
- Dicarboxylic acid or derivatives
- Pyran
- Monocyclic benzene moiety
- Benzenoid
- Carboxylic acid ester
- Secondary alcohol
- Polyol
- Ether
- Carboxylic acid
- Oxacycle
- Organoheterocyclic compound
- Carboxylic acid derivative
- Acetal
- Organooxygen compound
- Organic oxide
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Organic oxygen compound
- Carbonyl group
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0pbc-9154200000-8c81f08e6225a78fef0e | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-00xu-3073249000-153e2fd0b9e5dc827ea3 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0fkc-0960600000-04eb2a5669d2879077eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00dl-0980000000-1017b5d38cc4533d1dbd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05fr-0930000000-b43ee06cd57102bf87ce | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01bc-1696800000-df0f55939c39649542e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-1392000000-7069d48f679e01bf0e00 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01ox-3590000000-c75c55b408810c043444 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014j-0857900000-a950c090d318f318ae2e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000f-6394100000-df4ce18ed51b0ccd6305 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00kf-5970000000-7bb10f372be95fe1567f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0110900000-bdbeeb486d0753948b23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uy3-0339700000-0e609d9141104e6e072a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0g2a-1985300000-3728fb5b9dbd0b76e62b | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0041731 |
|---|
| FooDB ID | FDB029897 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30777624 |
|---|
| ChEBI ID | 176354 |
|---|
| PubChem Compound ID | 122400995 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Neveu V, Perez-Jimenez J, Vos F, Crespy V, du Chaffaut L, Mennen L, Knox C, Eisner R, Cruz J, Wishart D, Scalbert A: Phenol-Explorer: an online comprehensive database on polyphenol contents in foods. Database (Oxford). 2010;2010:bap024. doi: 10.1093/database/bap024. Epub 2010 Jan 8. |
|
|---|