| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:15:11 UTC |
|---|
| Update Date | 2016-11-09 01:22:24 UTC |
|---|
| Accession Number | CHEM041140 |
|---|
| Identification |
|---|
| Common Name | Alanyltryptophan |
|---|
| Class | Small Molecule |
|---|
| Description | Alanyltryptophan is a derivative of tryptophan. Tryptophan is an essential amino acid which is the precursor of serotonin. |
|---|
| Contaminant Sources | - FooDB Chemicals
- HMDB Contaminants - Urine
|
|---|
| Contaminant Type | Not Available |
|---|
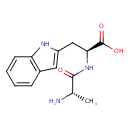
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ala-TRP | HMDB | | Alanyl tryptophan | HMDB | | L-Alanyl-L-tryptophan | HMDB | | L-Alanyltryptophan | HMDB |
|
|---|
| Chemical Formula | C14H17N3O3 |
|---|
| Average Molecular Mass | 275.303 g/mol |
|---|
| Monoisotopic Mass | 275.127 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2S)-2-[(2S)-2-aminopropanamido]-3-(1H-indol-2-yl)propanoic acid |
|---|
| Traditional Name | (2S)-2-[(2S)-2-aminopropanamido]-3-(1H-indol-2-yl)propanoic acid |
|---|
| SMILES | C[C@H](N)C(=O)N[C@@H](CC1=CC2=C(N1)C=CC=C2)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C14H17N3O3/c1-8(15)13(18)17-12(14(19)20)7-10-6-9-4-2-3-5-11(9)16-10/h2-6,8,12,16H,7,15H2,1H3,(H,17,18)(H,19,20)/t8-,12-/m0/s1 |
|---|
| InChI Key | CBUUWFVFZSZOAK-UFBFGSQYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as dipeptides. These are organic compounds containing a sequence of exactly two alpha-amino acids joined by a peptide bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Dipeptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-dipeptide
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- N-acyl-l-alpha-amino acid
- Alpha-amino acid amide
- Indolyl carboxylic acid derivative
- Alanine or derivatives
- Alpha-amino acid or derivatives
- Indole
- Indole or derivatives
- Benzenoid
- Substituted pyrrole
- Heteroaromatic compound
- Pyrrole
- Amino acid or derivatives
- Carboxamide group
- Amino acid
- Secondary carboxylic acid amide
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Azacycle
- Organoheterocyclic compound
- Organonitrogen compound
- Amine
- Organic oxide
- Primary aliphatic amine
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Organooxygen compound
- Primary amine
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0006-9220000000-27810ff95256929f8e9f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00dl-9201000000-9a6545288cd73aba99b6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-056r-3090000000-266fb47c9b0604d2c283 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-9320000000-77fef8fefa4cb2ec1484 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4l-7900000000-ae9b80fdb9db3c396e7a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0090000000-1951628148802deb47ea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0kni-3490000000-825d6f8f80b57608ceb6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05gu-9810000000-3bd30bf4c958b111dbd7 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0013209 |
|---|
| FooDB ID | FDB029335 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 53481655 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|