| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:10:26 UTC |
|---|
| Update Date | 2016-11-09 01:22:23 UTC |
|---|
| Accession Number | CHEM041052 |
|---|
| Identification |
|---|
| Common Name | Kinetensin |
|---|
| Class | Small Molecule |
|---|
| Description | An oligopeptide comprising of nine amino acids with sequence L-Ile-L-Ala-L-Arg-L-Arg-L-His-L-Pro-L-Tyr-L-Phe-L-Leu. It was originally isolated from pepsin-treated human plasma and shares some sequence homology with the C-terminal end of neurotensin. It is a potent histamine releaser and may serve as an inflammatory mediator. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
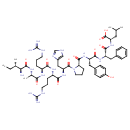
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| H-Ile-ala-arg-arg-his-pro-tyr-phe-leu-OH | ChEBI | | I-a-R-R-H-p-y-F-L | ChEBI | | IARRHPYFL | ChEBI | | Ile-ala-arg-arg-his-pro-tyr-phe-leu | ChEBI | | Kinetensin (human) | ChEBI | | L-Ile-L-ala-L-arg-L-arg-L-his-L-pro-L-tyr-L-phe-L-leu | ChEBI | | L-Isoleucyl-L-alanyl-L-arginyl-L-arginyl-L-histidyl-L-prolyl-L-tyrosyl-L-phenylalanyl-L-leucine | HMDB | | (2S)-2-{[(2S)-2-{[(2S)-2-({[(2S)-1-[(2S)-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-{[(2S,3S)-2-amino-1-hydroxy-3-methylpentylidene]amino}-1-hydroxypropylidene]amino}-5-carbamimidamido-1-hydroxypentylidene]amino}-5-carbamimidamido-1-hydroxypentylidene]amino}-3-(1H-imidazol-5-yl)propanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-1-hydroxy-3-(4-hydroxyphenyl)propylidene]amino}-1-hydroxy-3-phenylpropylidene]amino}-4-methylpentanoate | HMDB | | Kinetensin | MeSH |
|
|---|
| Chemical Formula | C56H85N17O11 |
|---|
| Average Molecular Mass | 1172.381 g/mol |
|---|
| Monoisotopic Mass | 1171.661 g/mol |
|---|
| CAS Registry Number | 103131-69-7 |
|---|
| IUPAC Name | (2S)-2-[(2S)-2-[(2S)-2-{[(2S)-1-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S,3S)-2-amino-3-methylpentanamido]propanamido]-5-carbamimidamidopentanamido]-5-carbamimidamidopentanamido]-3-(1H-imidazol-4-yl)propanoyl]pyrrolidin-2-yl]formamido}-3-(4-hydroxyphenyl)propanamido]-3-phenylpropanamido]-4-methylpentanoic acid |
|---|
| Traditional Name | (2S)-2-[(2S)-2-[(2S)-2-{[(2S)-1-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S,3S)-2-amino-3-methylpentanamido]propanamido]-5-carbamimidamidopentanamido]-5-carbamimidamidopentanamido]-3-(1H-imidazol-4-yl)propanoyl]pyrrolidin-2-yl]formamido}-3-(4-hydroxyphenyl)propanamido]-3-phenylpropanamido]-4-methylpentanoic acid |
|---|
| SMILES | CC[C@H](C)[C@H](N)C(=O)N[C@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CN=CN1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@H](CC1=CC=CC=C1)C(=O)N[C@H](CC(C)C)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C56H85N17O11/c1-6-32(4)45(57)52(81)66-33(5)46(75)67-38(15-10-22-63-55(58)59)47(76)68-39(16-11-23-64-56(60)61)48(77)71-42(28-36-29-62-30-65-36)53(82)73-24-12-17-44(73)51(80)70-41(27-35-18-20-37(74)21-19-35)49(78)69-40(26-34-13-8-7-9-14-34)50(79)72-43(54(83)84)25-31(2)3/h7-9,13-14,18-21,29-33,38-45,74H,6,10-12,15-17,22-28,57H2,1-5H3,(H,62,65)(H,66,81)(H,67,75)(H,68,76)(H,69,78)(H,70,80)(H,71,77)(H,72,79)(H,83,84)(H4,58,59,63)(H4,60,61,64)/t32-,33+,38-,39+,40+,41-,42-,43+,44-,45-/m0/s1 |
|---|
| InChI Key | PANUJGMSOSQAAY-HAGIGRARSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as oligopeptides. These are organic compounds containing a sequence of between three and ten alpha-amino acids joined by peptide bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Oligopeptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-oligopeptide
- Tyrosine or derivatives
- Phenylalanine or derivatives
- Histidine or derivatives
- Isoleucine or derivatives
- Leucine or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- N-acyl-l-alpha-amino acid
- Proline or derivatives
- Alpha-amino acid amide
- Alanine or derivatives
- Amphetamine or derivatives
- Alpha-amino acid or derivatives
- N-substituted-alpha-amino acid
- Pyrrolidine-2-carboxamide
- N-acylpyrrolidine
- Imidazolyl carboxylic acid derivative
- Pyrrolidine carboxylic acid or derivatives
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- N-acyl-amine
- Benzenoid
- Fatty acyl
- Fatty amide
- Monocyclic benzene moiety
- Imidazole
- Azole
- Tertiary carboxylic acid amide
- Pyrrolidine
- Heteroaromatic compound
- Amino acid or derivatives
- Amino acid
- Carboxamide group
- Guanidine
- Secondary carboxylic acid amide
- Propargyl-type 1,3-dipolar organic compound
- Monocarboxylic acid or derivatives
- Carboximidamide
- Organic 1,3-dipolar compound
- Organoheterocyclic compound
- Carboxylic acid
- Azacycle
- Organooxygen compound
- Amine
- Organopnictogen compound
- Carbonyl group
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Primary aliphatic amine
- Organic nitrogen compound
- Primary amine
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-052r-6900100010-fedb3c0f20f9a445f035 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-9600000000-ba6924fcc6755fbd5259 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0553-9400001000-bebff0501ef643c7cbd9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00b9-6900000000-2d29098a6602c501d22a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0563-9600111001-f9e131457a0b0975f16b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-055f-9310100211-585f8604e55684ae3196 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-1900000000-ce6e85c493a87521e48b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-9300000001-03a5ddb21811bf12eb72 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f96-9313010432-c55558c9dba01cb254cc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-05fr-7900000000-2d8c6727698732de7980 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0670-9000000001-c48ef271732fa0d837bd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-07xu-9200000000-b6db51da26c2a088ef1d | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0012988 |
|---|
| FooDB ID | FDB029235 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 129668 |
|---|
| ChEBI ID | 80144 |
|---|
| PubChem Compound ID | 147043 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|