| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:08:13 UTC |
|---|
| Update Date | 2016-11-09 01:22:23 UTC |
|---|
| Accession Number | CHEM041020 |
|---|
| Identification |
|---|
| Common Name | Apelin-13 |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
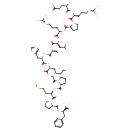
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Apelin-13 peptide | MeSH | | prepro-65-77-Apelin | HMDB | | (2R)-2-({[(2S)-1-[(2R)-2-({[(2R)-1-(2-{[(2R)-6-amino-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-({[(2R)-1-[(2S)-2-{[(2S)-2-amino-1-hydroxy-4-(C-hydroxycarbonimidoyl)butylidene]amino}-5-carbamimidamidopentanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-5-carbamimidamido-1-hydroxypentylidene]amino}-1-hydroxy-4-methylpentylidene]amino}-1,3-dihydroxypropylidene]amino}-1-hydroxy-3-(1H-imidazol-5-yl)propylidene]amino}-1-hydroxyhexylidene]amino}acetyl)pyrrolidin-2-yl](hydroxy)methylidene}amino)-4-(methylsulfanyl)butanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-3-phenylpropanoate | Generator | | (2R)-2-({[(2S)-1-[(2R)-2-({[(2R)-1-(2-{[(2R)-6-amino-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-({[(2R)-1-[(2S)-2-{[(2S)-2-amino-1-hydroxy-4-(C-hydroxycarbonimidoyl)butylidene]amino}-5-carbamimidamidopentanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-5-carbamimidamido-1-hydroxypentylidene]amino}-1-hydroxy-4-methylpentylidene]amino}-1,3-dihydroxypropylidene]amino}-1-hydroxy-3-(1H-imidazol-5-yl)propylidene]amino}-1-hydroxyhexylidene]amino}acetyl)pyrrolidin-2-yl](hydroxy)methylidene}amino)-4-(methylsulphanyl)butanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-3-phenylpropanoate | Generator | | (2R)-2-({[(2S)-1-[(2R)-2-({[(2R)-1-(2-{[(2R)-6-amino-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-{[(2S)-2-({[(2R)-1-[(2S)-2-{[(2S)-2-amino-1-hydroxy-4-(C-hydroxycarbonimidoyl)butylidene]amino}-5-carbamimidamidopentanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-5-carbamimidamido-1-hydroxypentylidene]amino}-1-hydroxy-4-methylpentylidene]amino}-1,3-dihydroxypropylidene]amino}-1-hydroxy-3-(1H-imidazol-5-yl)propylidene]amino}-1-hydroxyhexylidene]amino}acetyl)pyrrolidin-2-yl](hydroxy)methylidene}amino)-4-(methylsulphanyl)butanoyl]pyrrolidin-2-yl](hydroxy)methylidene}amino)-3-phenylpropanoic acid | Generator | | Apelin-13 | MeSH |
|
|---|
| Chemical Formula | C69H111N23O16S |
|---|
| Average Molecular Mass | 1550.829 g/mol |
|---|
| Monoisotopic Mass | 1549.830 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2R)-2-{[(2S)-1-[(2R)-2-{[(2R)-1-{2-[(2R)-6-amino-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-{[(2R)-1-[(2S)-2-[(2S)-2-amino-4-carbamoylbutanamido]-5-[(diaminomethylidene)amino]pentanoyl]pyrrolidin-2-yl]formamido}-5-[(diaminomethylidene)amino]pentanamido]-4-methylpentanamido]-3-hydroxypropanamido]-3-(1H-imidazol-5-yl)propanamido]hexanamido]acetyl}pyrrolidin-2-yl]formamido}-4-(methylsulfanyl)butanoyl]pyrrolidin-2-yl]formamido}-3-phenylpropanoic acid |
|---|
| Traditional Name | (2R)-2-{[(2S)-1-[(2R)-2-{[(2R)-1-{2-[(2R)-6-amino-2-[(2S)-2-[(2S)-2-[(2S)-2-[(2S)-2-{[(2R)-1-[(2S)-2-[(2S)-2-amino-4-carbamoylbutanamido]-5-[(diaminomethylidene)amino]pentanoyl]pyrrolidin-2-yl]formamido}-5-[(diaminomethylidene)amino]pentanamido]-4-methylpentanamido]-3-hydroxypropanamido]-3-(3H-imidazol-4-yl)propanamido]hexanamido]acetyl}pyrrolidin-2-yl]formamido}-4-(methylsulfanyl)butanoyl]pyrrolidin-2-yl]formamido}-3-phenylpropanoic acid |
|---|
| SMILES | CSCC[C@@H](NC(=O)[C@H]1CCCN1C(=O)CNC(=O)[C@@H](CCCCN)NC(=O)[C@H](CC1=CN=CN1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](N)CCC(N)=O)C(=O)N1CCC[C@H]1C(=O)N[C@H](CC1=CC=CC=C1)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C69H111N23O16S/c1-39(2)32-47(86-58(98)44(17-9-26-78-68(73)74)83-63(103)52-20-12-29-91(52)65(105)45(18-10-27-79-69(75)76)84-56(96)42(71)22-23-54(72)94)59(99)89-50(37-93)61(101)87-48(34-41-35-77-38-81-41)60(100)82-43(16-7-8-25-70)57(97)80-36-55(95)90-28-11-19-51(90)62(102)85-46(24-31-109-3)66(106)92-30-13-21-53(92)64(104)88-49(67(107)108)33-40-14-5-4-6-15-40/h4-6,14-15,35,38-39,42-53,93H,7-13,16-34,36-37,70-71H2,1-3H3,(H2,72,94)(H,77,81)(H,80,97)(H,82,100)(H,83,103)(H,84,96)(H,85,102)(H,86,98)(H,87,101)(H,88,104)(H,89,99)(H,107,108)(H4,73,74,78)(H4,75,76,79)/t42-,43+,44-,45-,46+,47-,48-,49+,50-,51+,52+,53-/m0/s1 |
|---|
| InChI Key | XXCCRHIAIBQDPX-YHQCEEEXSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as polypeptides. These are peptides containing ten or more amino acid residues. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic Polymers |
|---|
| Class | Polypeptides |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Polypeptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Polypeptide
- Alpha peptide
- Phenylalanine or derivatives
- Histidine or derivatives
- Glutamine or derivatives
- Methionine or derivatives
- Leucine or derivatives
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Proline or derivatives
- Serine or derivatives
- Alpha-amino acid amide
- 3-phenylpropanoic-acid
- Amphetamine or derivatives
- Alpha-amino acid or derivatives
- N-substituted-alpha-amino acid
- N-acylpyrrolidine
- Pyrrolidine carboxylic acid or derivatives
- Pyrrolidine-2-carboxamide
- Imidazolyl carboxylic acid derivative
- Benzenoid
- Fatty acyl
- Fatty amide
- Monocyclic benzene moiety
- N-acyl-amine
- Azole
- Pyrrolidine
- Tertiary carboxylic acid amide
- Heteroaromatic compound
- Imidazole
- Amino acid or derivatives
- Amino acid
- Carboxamide group
- Guanidine
- Secondary carboxylic acid amide
- Primary carboxylic acid amide
- Thioether
- Carboxylic acid derivative
- Carboxylic acid
- Azacycle
- Organoheterocyclic compound
- Dialkylthioether
- Monocarboxylic acid or derivatives
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Sulfenyl compound
- Carboximidamide
- Amine
- Primary alcohol
- Primary amine
- Organic oxygen compound
- Organopnictogen compound
- Alcohol
- Organosulfur compound
- Primary aliphatic amine
- Organic oxide
- Organic nitrogen compound
- Hydrocarbon derivative
- Carbonyl group
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0f89-2200090000-082156889447f3345908 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0f89-6922250020-31ec08ecb438cf19de98 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0v6u-9532200100-d80feb4bb0cf4ac7c780 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-06rj-3000890000-634f1d2b6dfd4644a8f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052k-9111640010-6124249b3e3cf13ec481 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0005-9202130011-7991b2aee185e1378dc9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000e-2010290001-8373f6245c7079b92dcb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01qd-5340491011-d9cf06507982d17caf9c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ufv-7802897263-f8e7ae4bd59f9f44cf98 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ue9-2011290011-fc0883ce78584d879019 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-2201091014-b2a33a4f2a10a06cda53 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03y0-9017072011-c67d52f4022ac3126ab9 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0012894 |
|---|
| FooDB ID | FDB029200 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30776660 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 53481538 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Zhou N, Fang J, Acheampong E, Mukhtar M, Pomerantz RJ: Binding of ALX40-4C to APJ, a CNS-based receptor, inhibits its utilization as a co-receptor by HIV-1. Virology. 2003 Jul 20;312(1):196-203. | | 2. Hashimoto Y, Ishida J, Yamamoto R, Fujiwara K, Asada S, Kasuya Y, Mochizuki N, Fukamizu A: G protein-coupled APJ receptor signaling induces focal adhesion formation and cell motility. Int J Mol Med. 2005 Nov;16(5):787-92. |
|
|---|