| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:07:55 UTC |
|---|
| Update Date | 2016-11-09 01:22:22 UTC |
|---|
| Accession Number | CHEM041008 |
|---|
| Identification |
|---|
| Common Name | 7'-Carboxy-gamma-tocotrienol |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
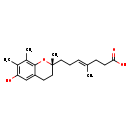
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 7'-Carboxy-g-tocotrienol | Generator | | 7'-Carboxy-γ-tocotrienol | Generator | | gamma-CMHenHC | HMDB | | γ-CMHenHC | HMDB | | γ-Carboxymethylhexenyl hydroxychroman | HMDB | | γ-Carboxymethylhexenylhydroxychroman | HMDB | | gamma-Carboxymethylhexenyl hydroxychroman | HMDB | | gamma-Carboxymethylhexenylhydroxychroman | HMDB | | (4E)-7-[(2R)-6-Hydroxy-2,7,8-trimethyl-3,4-dihydro-2H-1-benzopyran-2-yl]-4-methylhept-4-enoate | Generator |
|
|---|
| Chemical Formula | C20H28O4 |
|---|
| Average Molecular Mass | 332.434 g/mol |
|---|
| Monoisotopic Mass | 332.199 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (4E)-7-[(2R)-6-hydroxy-2,7,8-trimethyl-3,4-dihydro-2H-1-benzopyran-2-yl]-4-methylhept-4-enoic acid |
|---|
| Traditional Name | (4E)-7-[(2R)-6-hydroxy-2,7,8-trimethyl-3,4-dihydro-1-benzopyran-2-yl]-4-methylhept-4-enoic acid |
|---|
| SMILES | C\C(CCC(O)=O)=C/CC[C@]1(C)CCC2=C(O1)C(C)=C(C)C(O)=C2 |
|---|
| InChI Identifier | InChI=1S/C20H28O4/c1-13(7-8-18(22)23)6-5-10-20(4)11-9-16-12-17(21)14(2)15(3)19(16)24-20/h6,12,21H,5,7-11H2,1-4H3,(H,22,23)/b13-6+/t20-/m1/s1 |
|---|
| InChI Key | NECINJBBPZOJSN-QARGNGGGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 1-benzopyrans. These are organic aromatic compounds that 1-benzopyran, a bicyclic compound made up of a benzene ring fused to a pyran, so that the oxygen atom is at the 1-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzopyrans |
|---|
| Sub Class | 1-benzopyrans |
|---|
| Direct Parent | 1-benzopyrans |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1-benzopyran
- Medium-chain fatty acid
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Branched fatty acid
- Heterocyclic fatty acid
- Hydroxy fatty acid
- Methyl-branched fatty acid
- Unsaturated fatty acid
- Fatty acyl
- Fatty acid
- Benzenoid
- Monocarboxylic acid or derivatives
- Oxacycle
- Ether
- Carboxylic acid
- Carboxylic acid derivative
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Carbonyl group
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00kf-6793000000-1f4f7e996607646580d8 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-03ki-9226700000-4ad9ef42d5fed71097e4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0539000000-c24b18697e6615eaac51 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-1911000000-0757b6d1d9deb8db59da | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0uxr-5900000000-c32bf0a50ecc39c82f83 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0119000000-dc6e16f6c4bfd399878a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01pt-1749000000-1d14d6e939c0373bc7db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4j-9731000000-5018e8a422c062384d6d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0019-0097000000-94c5e80f1049b5b1e13a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06uv-5539000000-6783a4bcc672226a5153 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-009m-2930000000-a5afd52373b933a4ada7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0159-0296000000-43779c4e87162f7210cb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001v-2490000000-6ff63b8a8fc2ee13c47b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ffc-5900000000-17f034b4bd2d114161fd | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0012851 |
|---|
| FooDB ID | FDB029188 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30776648 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 53481532 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Birringer M, Pfluger P, Kluth D, Landes N, Brigelius-Flohe R: Identities and differences in the metabolism of tocotrienols and tocopherols in HepG2 cells. J Nutr. 2002 Oct;132(10):3113-8. | | 2. Zhao Y, Lee MJ, Cheung C, Ju JH, Chen YK, Liu B, Hu LQ, Yang CS: Analysis of multiple metabolites of tocopherols and tocotrienols in mice and humans. J Agric Food Chem. 2010 Apr 28;58(8):4844-52. doi: 10.1021/jf904464u. |
|
|---|