| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:07:54 UTC |
|---|
| Update Date | 2016-11-09 01:22:22 UTC |
|---|
| Accession Number | CHEM041007 |
|---|
| Identification |
|---|
| Common Name | 7'-Carboxy-gamma-chromanol |
|---|
| Class | Small Molecule |
|---|
| Description | 7'-Carboxy-gamma-tocopherol is a dehydrogenation carboxylate product of 7'-hydroxy-r-tocopherol by an unidentified microsomal enzyme(s) probably via an aldehyde intermediate. r-Tocopherol provides different antioxidant activities in food and in-vitro studies and showed higher activity in trapping lipophilic electrophiles and reactive nitrogen and oxygen species. From the metabolism end product, only that of r-tocopherol (2,7,8-trimethyl-2-(b-carboxyethyl)-6-hydroxychroman), but not that of a-tocopherol, was identified to provide natriuretic activity. Only the r-tocopherol plasma level served as biomarker for cancer and cardiovascular risk. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
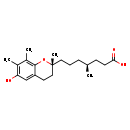
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (4S)-7-[(2R)-6-Hydroxy-2,7,8-trimethyl-3,4-dihydro-2H-1-benzopyran-2-yl]-4-methylheptanoate | HMDB | | Γ-CMHHC | HMDB | | gamma-CMHHC | HMDB | | γ-Carboxymethylhexyl hydroxychroman | HMDB | | γ-Carboxymethylhexylhydroxychroman | HMDB | | gamma-Carboxymethylhexyl hydroxychroman | HMDB | | gamma-Carboxymethylhexylhydroxychroman | HMDB |
|
|---|
| Chemical Formula | C20H30O4 |
|---|
| Average Molecular Mass | 334.450 g/mol |
|---|
| Monoisotopic Mass | 334.214 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (4S)-7-[(2R)-6-hydroxy-2,7,8-trimethyl-3,4-dihydro-2H-1-benzopyran-2-yl]-4-methylheptanoic acid |
|---|
| Traditional Name | (4S)-7-[(2R)-6-hydroxy-2,7,8-trimethyl-3,4-dihydro-1-benzopyran-2-yl]-4-methylheptanoic acid |
|---|
| SMILES | C[C@H](CCC[C@]1(C)CCC2=C(O1)C(C)=C(C)C(O)=C2)CCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H30O4/c1-13(7-8-18(22)23)6-5-10-20(4)11-9-16-12-17(21)14(2)15(3)19(16)24-20/h12-13,21H,5-11H2,1-4H3,(H,22,23)/t13-,20-/m1/s1 |
|---|
| InChI Key | JFXDIXKMINYLIK-ZUOKHONESA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 1-benzopyrans. These are organic aromatic compounds that 1-benzopyran, a bicyclic compound made up of a benzene ring fused to a pyran, so that the oxygen atom is at the 1-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzopyrans |
|---|
| Sub Class | 1-benzopyrans |
|---|
| Direct Parent | 1-benzopyrans |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1-benzopyran
- Medium-chain fatty acid
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Branched fatty acid
- Heterocyclic fatty acid
- Hydroxy fatty acid
- Methyl-branched fatty acid
- Fatty acyl
- Fatty acid
- Benzenoid
- Monocarboxylic acid or derivatives
- Oxacycle
- Ether
- Carboxylic acid
- Carboxylic acid derivative
- Organooxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic oxygen compound
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014r-0539000000-6050268e609a16466634 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-1921000000-7fda212da468ee981e95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-1000-5900000000-5c8150efaf5eae1bce1f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0119000000-9efcbf3debf6e3fab178 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00lb-1749000000-0ef6f3c235f3a19bf981 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4j-9841000000-7e52b90564e14a9bfb9e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00li-0059000000-951a5f97c523abed2dbc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00kb-2936000000-ad8508fe5d137c7c62b0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0076-2951000000-edc264cfd7d7cc48b18b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00ks-0195000000-f475a6e7b1f959cd7904 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-1390000000-abe1846fbee337e380b1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fdo-5900000000-892525c38c02793675e1 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0012850 |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 74854294 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 134159053 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|