| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-27 01:07:34 UTC |
|---|
| Update Date | 2016-11-09 01:22:22 UTC |
|---|
| Accession Number | CHEM040993 |
|---|
| Identification |
|---|
| Common Name | 4-Methoxy-17beta-estradiol |
|---|
| Class | Small Molecule |
|---|
| Description | A 17beta-hydroxy steroid that is 17beta-estradiol in which the hydrogen at position 4 has been replaced by a methoxy group. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
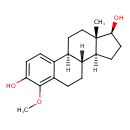
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (17beta)-4-Methoxyestra-1,3,5(10)-triene-3,17-diol | ChEBI | | 4-MeOE2 | ChEBI | | 4-Methoxy-3,17beta-dihydroxy-1,3,5[10]-estratriene | ChEBI | | 4-Methoxyestradiol | ChEBI | | 4-Methoxyestradiol-17beta | ChEBI | | (17b)-4-Methoxyestra-1,3,5(10)-triene-3,17-diol | Generator | | (17Β)-4-methoxyestra-1,3,5(10)-triene-3,17-diol | Generator | | 4-Methoxy-3,17b-dihydroxy-1,3,5[10]-estratriene | Generator | | 4-Methoxy-3,17β-dihydroxy-1,3,5[10]-estratriene | Generator | | 4-Methoxyestradiol-17b | Generator | | 4-Methoxyestradiol-17β | Generator | | 4-Methoxy-17b-estradiol | Generator | | 4-Methoxy-17β-estradiol | Generator | | 3,17beta-Dihydroxy-4-methoxy-1,3,5[10]-estratriene | HMDB | | 3,4,17beta-Trihydroxy-1,3,5[10]-estratriene 4-methyl ether | HMDB | | 4-Methoxy-1,3,5[10]-estratriene-3,17beta-diol | HMDB | | 4-Methoxyestradiol-17 beta | HMDB | | 4-ME2 | HMDB |
|
|---|
| Chemical Formula | C19H26O3 |
|---|
| Average Molecular Mass | 302.408 g/mol |
|---|
| Monoisotopic Mass | 302.188 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (1S,10R,11S,14S,15S)-6-methoxy-15-methyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-2,4,6-triene-5,14-diol |
|---|
| Traditional Name | (1S,10R,11S,14S,15S)-6-methoxy-15-methyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-2,4,6-triene-5,14-diol |
|---|
| SMILES | COC1=C2CCC3C4CC[C@H](O)[C@@]4(C)CCC3C2=CC=C1O |
|---|
| InChI Identifier | InChI=1S/C19H26O3/c1-19-10-9-12-11-5-7-16(20)18(22-2)14(11)4-3-13(12)15(19)6-8-17(19)21/h5,7,12-13,15,17,20-21H,3-4,6,8-10H2,1-2H3/t12?,13?,15?,17-,19-/m0/s1 |
|---|
| InChI Key | BCWZIZLVBYHFES-BHFCKAAKSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as estrogens and derivatives. These are steroids with a structure containing a 3-hydroxylated estrane. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Estrane steroids |
|---|
| Direct Parent | Estrogens and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Estrogen-skeleton
- 3-hydroxysteroid
- 17-hydroxysteroid
- Hydroxysteroid
- Phenanthrene
- Tetralin
- Anisole
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Benzenoid
- Cyclic alcohol
- Secondary alcohol
- Ether
- Organic oxygen compound
- Organooxygen compound
- Hydrocarbon derivative
- Alcohol
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0f79-0097000000-571b294f162709cb964d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0f79-0692000000-65b75c1a518e2e6a402b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000l-5590000000-885e6b66b6f87173aa71 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0019000000-2e5aedfaa3bc522112af | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0069000000-2106698fbc9b5342a9b1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a73-1090000000-d629405cab5cc07c6d48 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0029000000-3f80ac8b07ab8f4dd0ff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0zg0-0983000000-0faf51d5791a2d67d37a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a70-1920000000-906d2e9b3ff38c06aca3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0009000000-4360bacaf95498925fe5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0019000000-08de12b7155407976f40 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0gb9-0090000000-39ecefcaad2cacbbe957 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0012782 |
|---|
| FooDB ID | FDB029173 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 61848 |
|---|
| ChEBI ID | 136975 |
|---|
| PubChem Compound ID | 68578 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|