| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 15:19:25 UTC |
|---|
| Update Date | 2016-11-09 01:21:59 UTC |
|---|
| Accession Number | CHEM039057 |
|---|
| Identification |
|---|
| Common Name | PE(24:0/24:0) |
|---|
| Class | Small Molecule |
|---|
| Description | PE(24:0/24:0), also known as GPEtn(48:0), belongs to the class of organic compounds known as phosphatidylethanolamines. These are glycerophosphoetahnolamines in which two fatty acids are bonded to the glycerol moiety through ester linkages. PE(24:0/24:0) is possibly soluble (in water) and a very strong basic compound (based on its pKa). PE(24:0/24:0) participates in a number of enzymatic reactions, within cattle. In particular, Cytidine monophosphate and PE(24:0/24:0) can be biosynthesized from CDP-ethanolamine and DG(24:0/24:0/0:0); which is catalyzed by the enzyme choline/ethanolaminephosphotransferase. Furthermore, PE(24:0/24:0) can be biosynthesized from PS(24:0/24:0); which is catalyzed by the enzyme phosphatidylserine decarboxylase. Furthermore, PE(24:0/24:0) can be biosynthesized from PS(24:0/24:0); which is mediated by the enzyme phosphatidylserine decarboxylase. Finally, Cytidine monophosphate and PE(24:0/24:0) can be biosynthesized from CDP-ethanolamine and DG(24:0/24:0/0:0) through its interaction with the enzyme choline/ethanolaminephosphotransferase. In cattle, PE(24:0/24:0) is involved in a couple of metabolic pathways, which include phosphatidylcholine biosynthesis PC(24:0/24:0) pathway and phosphatidylethanolamine biosynthesis pe(24:0/24:0) pathway. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
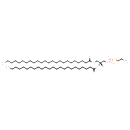
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,2-dilignoceroyl-rac-glycero-3-phosphoethanolamine | Lipid Annotator, HMDB | | PE(24:0/24:0) | Lipid Annotator | | Phophatidylethanolamine(24:0/24:0) | Lipid Annotator, HMDB | | GPEtn(24:0/24:0) | Lipid Annotator, HMDB | | GPEtn(48:0) | Lipid Annotator, HMDB | | Phophatidylethanolamine(48:0) | Lipid Annotator, HMDB | | 1,2-ditetracosanoyl-rac-glycero-3-phosphoethanolamine | Lipid Annotator, HMDB | | PE(48:0) | Lipid Annotator, HMDB |
|
|---|
| Chemical Formula | C53H106NO8P |
|---|
| Average Molecular Mass | 916.384 g/mol |
|---|
| Monoisotopic Mass | 915.766 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2-aminoethoxy)[(2R)-2,3-bis(tetracosanoyloxy)propoxy]phosphinic acid |
|---|
| Traditional Name | 2-aminoethoxy(2R)-2,3-bis(tetracosanoyloxy)propoxyphosphinic acid |
|---|
| SMILES | [H][C@@](COC(=O)CCCCCCCCCCCCCCCCCCCCCCC)(COP(O)(=O)OCCN)OC(=O)CCCCCCCCCCCCCCCCCCCCCCC |
|---|
| InChI Identifier | InChI=1S/C53H106NO8P/c1-3-5-7-9-11-13-15-17-19-21-23-25-27-29-31-33-35-37-39-41-43-45-52(55)59-49-51(50-61-63(57,58)60-48-47-54)62-53(56)46-44-42-40-38-36-34-32-30-28-26-24-22-20-18-16-14-12-10-8-6-4-2/h51H,3-50,54H2,1-2H3,(H,57,58)/t51-/m1/s1 |
|---|
| InChI Key | BZUNRSOACSOEBT-NLXJDERGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phosphatidylethanolamines. These are glycerophosphoetahnolamines in which two fatty acids are bonded to the glycerol moiety through ester linkages. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Glycerophospholipids |
|---|
| Sub Class | Glycerophosphoethanolamines |
|---|
| Direct Parent | Phosphatidylethanolamines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diacylglycero-3-phosphoethanolamine
- Phosphoethanolamine
- Fatty acid ester
- Dialkyl phosphate
- Dicarboxylic acid or derivatives
- Organic phosphoric acid derivative
- Phosphoric acid ester
- Alkyl phosphate
- Fatty acyl
- Amino acid or derivatives
- Carboxylic acid ester
- Carboxylic acid derivative
- Organopnictogen compound
- Organic oxygen compound
- Organooxygen compound
- Organonitrogen compound
- Amine
- Primary aliphatic amine
- Organic nitrogen compound
- Primary amine
- Carbonyl group
- Organic oxide
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0001000009-a480fee3380634b8fe5c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0001000009-a480fee3380634b8fe5c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-02t9-0309030006-9c03f4a76a803decf7db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0001000009-24071adeb300f4d3f7df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0001000009-24071adeb300f4d3f7df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-02t9-0309030006-08311ae0361fbc5ee1d2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000000119-ae6ab05def47bddf0a68 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00or-0000130916-01a245b542c101e8dbb3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-0000130911-71ce4ac87e151f6df056 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000000119-617cc6df0b8c18dc33d4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00or-0000130916-fbec4b1340f056fd54db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-0000130911-80020ece3d684482e9ba | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0000000109-d0d246d7c6f581226e50 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000j-0000000229-8a3e9ce70715772f1a39 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-0100010191-fee81e29782720991af2 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0009739 |
|---|
| FooDB ID | FDB026929 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 24769179 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 53480011 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|