| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 07:22:01 UTC |
|---|
| Update Date | 2016-11-09 01:21:28 UTC |
|---|
| Accession Number | CHEM036309 |
|---|
| Identification |
|---|
| Common Name | 2-Hydroxy-3-(4-hydroxyphenyl)propenoic acid |
|---|
| Class | Small Molecule |
|---|
| Description | A 2-hydroxy monocarboxylic acid that is acrylic acid in which the hydrogen at position 2 is substituted by a hydroxy group and a hydrogen at position 3 is substituted by a 4-hydroxyphenyl group. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
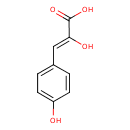
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4,alpha-Dihydroxycinnamic acid | ChEBI | | 4-Hydroxy-enol-phenylpyruvate | Kegg | | 4,a-Dihydroxycinnamate | Generator | | 4,a-Dihydroxycinnamic acid | Generator | | 4,alpha-Dihydroxycinnamate | Generator | | 4,Α-dihydroxycinnamate | Generator | | 4,Α-dihydroxycinnamic acid | Generator | | 4-Hydroxy-enol-phenylpyruvic acid | Generator | | 2-Hydroxy-3-(4-hydroxyphenyl)propenoate | Generator |
|
|---|
| Chemical Formula | C9H8O4 |
|---|
| Average Molecular Mass | 180.157 g/mol |
|---|
| Monoisotopic Mass | 180.042 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2Z)-2-hydroxy-3-(4-hydroxyphenyl)prop-2-enoic acid |
|---|
| Traditional Name | 4,α-dihydroxycinnamic acid |
|---|
| SMILES | OC(=O)C(\O)=C\C1=CC=C(O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C9H8O4/c10-7-3-1-6(2-4-7)5-8(11)9(12)13/h1-5,10-11H,(H,12,13)/b8-5- |
|---|
| InChI Key | GQYBCIHRWMPOOF-YVMONPNESA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylpyruvic acid derivatives. Phenylpyruvic acid derivatives are compounds containing a phenylpyruvic acid moiety, which consists of a phenyl group substituted at the second position by an pyruvic acid. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Phenylpyruvic acid derivatives |
|---|
| Direct Parent | Phenylpyruvic acid derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cinnamic acid
- Cinnamic acid or derivatives
- Coumaric acid
- Coumaric acid or derivatives
- Hydroxycinnamic acid
- Hydroxycinnamic acid or derivatives
- Enol-phenylpyruvate
- Phenol
- 1-hydroxy-2-unsubstituted benzenoid
- Monocarboxylic acid or derivatives
- Enol
- Carboxylic acid
- Carboxylic acid derivative
- Carbonyl group
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-053r-1900000000-10c51d0329cb2a35dbb8 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-00gi-6259000000-cdf2744454615081e02f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0bu0-0900000000-a08730e02c84f43e7926 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0900000000-2eb4f2eaa5d547249a3e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-7900000000-63f2942443ac7bfdce48 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0900000000-8576bdecf0b58e989df6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-0900000000-46e515a63da5ddf92de9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0900000000-3c0a27f480b5bb59093c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0900000000-02f0fa9241cf4b1591c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-9800000000-0e7b12140d77aa62a512 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0api-4900000000-6f21f2ecdec85109e7d5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01qi-0900000000-28cf4812bb35ce056a15 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-1900000000-bd40f97e341181046c0f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-9200000000-c7f40face58730829b4a | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0006915 |
|---|
| FooDB ID | FDB024152 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 552441 |
|---|
| ChEBI ID | 27683 |
|---|
| PubChem Compound ID | 636708 |
|---|
| Kegg Compound ID | C05350 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | ECMDB24915 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|