| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 07:13:47 UTC |
|---|
| Update Date | 2016-11-09 01:21:26 UTC |
|---|
| Accession Number | CHEM036085 |
|---|
| Identification |
|---|
| Common Name | cis-2-Methylaconitate |
|---|
| Class | Small Molecule |
|---|
| Description | A tricarboxylic acid comprising (Z)-but-2-ene having the three carboxy groups at the 1-, 2- and 3-positions. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
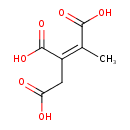
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (Z)-But-2-ene-1,2,3-tricarboxylate | ChEBI | | alpha-Methyl-cis-aconitate | ChEBI | | alpha-Methylaconitate | ChEBI | | cis-2-Butene-1,2,3-tricarboxylic acid | ChEBI | | (Z)-But-2-ene-1,2,3-tricarboxylic acid | Generator | | a-Methyl-cis-aconitate | Generator | | a-Methyl-cis-aconitic acid | Generator | | alpha-Methyl-cis-aconitic acid | Generator | | Α-methyl-cis-aconitate | Generator | | Α-methyl-cis-aconitic acid | Generator | | a-Methylaconitate | Generator | | a-Methylaconitic acid | Generator | | alpha-Methylaconitic acid | Generator | | Α-methylaconitate | Generator | | Α-methylaconitic acid | Generator | | cis-2-Butene-1,2,3-tricarboxylate | Generator | | cis-2-Methylaconitic acid | Generator | | (2Z)-But-2-ene-1,2,3-tricarboxylic acid | HMDB | | (Z)-2-Butene-1,2,3-tricarboxylic acid | HMDB |
|
|---|
| Chemical Formula | C7H8O6 |
|---|
| Average Molecular Mass | 188.135 g/mol |
|---|
| Monoisotopic Mass | 188.032 g/mol |
|---|
| CAS Registry Number | 6061-93-4 |
|---|
| IUPAC Name | (1Z)-1-methylprop-1-ene-1,2,3-tricarboxylic acid |
|---|
| Traditional Name | α-methylaconitate |
|---|
| SMILES | C\C(C(O)=O)=C(/CC(O)=O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C7H8O6/c1-3(6(10)11)4(7(12)13)2-5(8)9/h2H2,1H3,(H,8,9)(H,10,11)(H,12,13)/b4-3- |
|---|
| InChI Key | NUZLRKBHOBPTQV-ARJAWSKDSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as tricarboxylic acids and derivatives. These are carboxylic acids containing exactly three carboxyl groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Tricarboxylic acids and derivatives |
|---|
| Direct Parent | Tricarboxylic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tricarboxylic acid or derivatives
- Carboxylic acid
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-002g-6900000000-ffb445dd87ee618648ff | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-0079-9388000000-0ff4121be9b872f0c1ab | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00di-1900000000-202d4666a34f5b4f09f9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00fs-5900000000-9c256387d352a7a6ebb2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-002b-9500000000-027a2750edf1831b7088 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000f-1900000000-e925a682247370a2fa7f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00mn-4900000000-ec6a931ab7e2e1653ea8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00dj-9200000000-07d3197108036a4cb0ff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-002e-4900000000-fe6584e3d383728ee001 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-9200000000-80caf61386c108fcd0b4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0002-9100000000-327d1a78edc99f8fe760 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0fi0-0900000000-734e75ff99c6e837cdcb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-002b-9600000000-065cce37ffa797ab854c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-006t-9000000000-925f178b16f34b5de908 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0006357 |
|---|
| FooDB ID | FDB023904 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | 43396 |
|---|
| BioCyc ID | CPD-1136 |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 2338376 |
|---|
| ChEBI ID | 16717 |
|---|
| PubChem Compound ID | 3080625 |
|---|
| Kegg Compound ID | C04225 |
|---|
| YMDB ID | YMDB00380 |
|---|
| ECMDB ID | ECMDB06357 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|