| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 07:10:44 UTC |
|---|
| Update Date | 2016-11-09 01:21:25 UTC |
|---|
| Accession Number | CHEM036048 |
|---|
| Identification |
|---|
| Common Name | 3,4-Dihydroxymandelaldehyde |
|---|
| Class | Small Molecule |
|---|
| Description | 3,4-Dihydroxymandelaldehyde, also known as 3,4-dihydroxymandelaldehyde or 3,4-dihydroxymandelaldehyde, belongs to the class of organic compounds known as phenylacetaldehydes. Phenylacetaldehydes are compounds containing a phenylacetaldehyde moiety, which consists of a phenyl group substituted at the second position by an acetalydehyde. 3,4-Dihydroxymandelaldehyde is possibly soluble (in water) and an extremely weak basic (essentially neutral) compound (based on its pKa). 3,4-Dihydroxymandelaldehyde exists in all living organisms, ranging from bacteria to humans. 3,4-Dihydroxymandelaldehyde participates in a number of enzymatic reactions, within cattle. In particular, 3,4-Dihydroxymandelaldehyde can be biosynthesized from norepinephrine through the action of the enzyme amine oxidase [flavin-containing] a. In addition, 3,4-Dihydroxymandelaldehyde and methylamine can be biosynthesized from epinephrine; which is mediated by the enzyme amine oxidase [flavin-containing] a. In cattle, 3,4-dihydroxymandelaldehyde is involved in the metabolic pathway called the tyrosine metabolism pathway. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
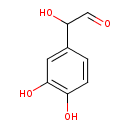
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3,4-Dihydroxymandelic aldehyde | ChEBI | | 3,4-Dihydroxyphenylglycolaldehyde | ChEBI | | 3,4-Dihydroxyphenylglycolic aldehyde | ChEBI | | alpha,3,4-Trihydroxybenzeneacetaldehyde | ChEBI | | alpha,3,4-Trihydroxyphenylacetaldehyde | ChEBI | | DHMAL | ChEBI | | DHPGALD | ChEBI | | DOPEGAL | ChEBI | | a,3,4-Trihydroxybenzeneacetaldehyde | Generator | | Α,3,4-trihydroxybenzeneacetaldehyde | Generator | | a,3,4-Trihydroxyphenylacetaldehyde | Generator | | Α,3,4-trihydroxyphenylacetaldehyde | Generator | | a,3,4-Trihydroxy-benzeneacetaldehyde | HMDB | | alpha,3,4-Trihydroxy-benzeneacetaldehyde | HMDB |
|
|---|
| Chemical Formula | C8H8O4 |
|---|
| Average Molecular Mass | 168.147 g/mol |
|---|
| Monoisotopic Mass | 168.042 g/mol |
|---|
| CAS Registry Number | 13023-73-9 |
|---|
| IUPAC Name | 2-(3,4-dihydroxyphenyl)-2-hydroxyacetaldehyde |
|---|
| Traditional Name | dhmal |
|---|
| SMILES | OC(C=O)C1=CC(O)=C(O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C8H8O4/c9-4-8(12)5-1-2-6(10)7(11)3-5/h1-4,8,10-12H |
|---|
| InChI Key | YUGMCLJIWGEKCK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as phenylacetaldehydes. Phenylacetaldehydes are compounds containing a phenylacetaldehyde moiety, which consists of a phenyl group substituted at the second position by an acetalydehyde. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Phenylacetaldehydes |
|---|
| Direct Parent | Phenylacetaldehydes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Phenylacetaldehyde
- Catechol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Alpha-hydroxyaldehyde
- Secondary alcohol
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organooxygen compound
- Organic oxygen compound
- Carbonyl group
- Aldehyde
- Aromatic alcohol
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-06ri-1900000000-d14339278a251a7d9df1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-01b9-7079000000-441c5f66bee2b8f14be7 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0900000000-01c8f9ca75c86beb7118 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0gb9-0900000000-30165da526b8042721a9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0kn9-9800000000-24ef8505a6b8f892128d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0900000000-2ce2cd9a3f1a5d4737b3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0aor-0900000000-a9b6cf257bb2c852ffd9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-6900000000-1f2d65b1c1a32c4ea1fd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014j-0900000000-876434025a3e5951556f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-0900000000-667ec91845715350734f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9200000000-1663e47bc16a5fa465e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0fki-0900000000-d67f635e503103dc887e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0r7i-4900000000-0f535b7035498a1a6303 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ue9-9200000000-344fc9120de4f34191d3 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0006242 |
|---|
| FooDB ID | FDB030386 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | 46056 |
|---|
| BioCyc ID | DIHYDROXYPHENYLGLYCOLALDEHYDE |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 133725 |
|---|
| ChEBI ID | 27852 |
|---|
| PubChem Compound ID | 151725 |
|---|
| Kegg Compound ID | C05577 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | ECMDB06242 |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Marchitti SA, Deitrich RA, Vasiliou V: Neurotoxicity and metabolism of the catecholamine-derived 3,4-dihydroxyphenylacetaldehyde and 3,4-dihydroxyphenylglycolaldehyde: the role of aldehyde dehydrogenase. Pharmacol Rev. 2007 Jun;59(2):125-50. Epub 2007 Mar 22. | | 2. Burke WJ, Li SW, Chung HD, Ruggiero DA, Kristal BS, Johnson EM, Lampe P, Kumar VB, Franko M, Williams EA, Zahm DS: Neurotoxicity of MAO metabolites of catecholamine neurotransmitters: role in neurodegenerative diseases. Neurotoxicology. 2004 Jan;25(1-2):101-15. | | 3. Burke WJ, Li SW, Zahm DS, Macarthur H, Kolo LL, Westfall TC, Anwar M, Glickstein SB, Ruggiero DA: Catecholamine monoamine oxidase a metabolite in adrenergic neurons is cytotoxic in vivo. Brain Res. 2001 Feb 9;891(1-2):218-27. | | 4. https://www.ncbi.nlm.nih.gov/pubmed/?term=10424772 | | 5. https://www.ncbi.nlm.nih.gov/pubmed/?term=11532332 | | 6. https://www.ncbi.nlm.nih.gov/pubmed/?term=12729575 | | 7. https://www.ncbi.nlm.nih.gov/pubmed/?term=14697885 | | 8. https://www.ncbi.nlm.nih.gov/pubmed/?term=18783367 | | 9. https://www.ncbi.nlm.nih.gov/pubmed/?term=8813375 | | 10. https://www.ncbi.nlm.nih.gov/pubmed/?term=9237550 | | 11. https://www.ncbi.nlm.nih.gov/pubmed/?term=9518674 | | 12. https://www.ncbi.nlm.nih.gov/pubmed/?term=9878889 |
|
|---|