| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 06:43:40 UTC |
|---|
| Update Date | 2016-11-09 01:21:23 UTC |
|---|
| Accession Number | CHEM035859 |
|---|
| Identification |
|---|
| Common Name | Thromboxane B3 |
|---|
| Class | Small Molecule |
|---|
| Description | |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
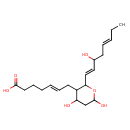
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (5Z,9a,13E,15S,17Z)-9,11,15-Trihydroxy-thromboxa-5,13,17-trien-1-Oate | HMDB | | (5Z,9a,13E,15S,17Z)-9,11,15-Trihydroxy-thromboxa-5,13,17-trien-1-Oic acid | HMDB | | 9S,11,15S-Trihydroxy-thromboxa-5Z,13E,17Z-trien-1-Oate | HMDB | | 9S,11,15S-Trihydroxy-thromboxa-5Z,13E,17Z-trien-1-Oic acid | HMDB | | TXB3 | HMDB, MeSH | | [2R(1E,3S,5Z),3S(Z),4S]-[Partial]-7-[tetrahydro-4,6-dihydroxy-2-(3-hydroxy-1,5-octadienyl)-2H-pyran-3-yl]-5-heptenoate | HMDB | | [2R(1E,3S,5Z),3S(Z),4S]-[Partial]-7-[tetrahydro-4,6-dihydroxy-2-(3-hydroxy-1,5-octadienyl)-2H-pyran-3-yl]-5-heptenoic acid | HMDB |
|
|---|
| Chemical Formula | C20H32O6 |
|---|
| Average Molecular Mass | 368.465 g/mol |
|---|
| Monoisotopic Mass | 368.220 g/mol |
|---|
| CAS Registry Number | 71953-80-5 |
|---|
| IUPAC Name | (5E)-7-{4,6-dihydroxy-2-[(1E,5E)-3-hydroxyocta-1,5-dien-1-yl]oxan-3-yl}hept-5-enoic acid |
|---|
| Traditional Name | thromboxane B3 |
|---|
| SMILES | CC\C=C\CC(O)\C=C\C1OC(O)CC(O)C1C\C=C\CCCC(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H32O6/c1-2-3-6-9-15(21)12-13-18-16(17(22)14-20(25)26-18)10-7-4-5-8-11-19(23)24/h3-4,6-7,12-13,15-18,20-22,25H,2,5,8-11,14H2,1H3,(H,23,24)/b6-3+,7-4+,13-12+ |
|---|
| InChI Key | OYPPJMLKAYYWHH-OHVJZDGFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as thromboxanes. These are eicosanoids structurally characterized by the presence of a 6-member ether containing ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Eicosanoids |
|---|
| Direct Parent | Thromboxanes |
|---|
| Alternative Parents | |
|---|
| Substituents | - Thromboxane
- Long-chain fatty acid
- Heterocyclic fatty acid
- Hydroxy fatty acid
- Fatty acid
- Oxane
- Unsaturated fatty acid
- Hemiacetal
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Oxacycle
- Monocarboxylic acid or derivatives
- Organoheterocyclic compound
- Organic oxide
- Organic oxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Alcohol
- Organooxygen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0f6x-9667000000-f4ce2aa3c5ab9901d9ba | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (4 TMS) - 70eV, Positive | splash10-0006-4300089000-51bd4164d954c93c28e4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0uei-0239000000-d3055e5b46f3ead5d5ea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0pc0-2149000000-3a365f4768659805dca5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014r-9210000000-6b4c19a67971e21744af | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-016v-0059000000-f381f931b44061fd5515 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-2579000000-1c860ae580e6e8ed5349 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4m-9551000000-d08a5187728b44f9f928 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-5a2cdf7255286b655e61 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-066s-1069000000-69f11b87e1bf34bbe824 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0007-7390000000-b258f7d4798fa158b519 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ue9-0019000000-9a935972b1a88c20ecaa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-053u-9313000000-663c96f23d66b2d8a26d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0api-9410000000-464dcb232bc3855b4826 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0005099 |
|---|
| FooDB ID | FDB023630 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4943166 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 6438711 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|