| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 05:54:39 UTC |
|---|
| Update Date | 2016-11-09 01:21:20 UTC |
|---|
| Accession Number | CHEM035592 |
|---|
| Identification |
|---|
| Common Name | (S)-Hydroxyoctanoyl-CoA |
|---|
| Class | Small Molecule |
|---|
| Description | Coenzyme A is notable for its role in the synthesis and oxidation of fatty acids. Since coenzyme A is chemically a thiol, it can react with carboxylic acids to form thioesters, thus functioning as an acyl group carrier. It assists in transferring fatty acids from the cytoplasm to mitochondria. Specifically (S)-Hydroxyoctanoyl-CoA is involved in fatty acid metabolism. It is the product of a reaction between 3-Oxooctanoyl-CoA and two enzymes; 3-hydroxyacyl-CoA Dehydrogenase and long-chain- 3-hydroxyacyl-CoA dehydrogenase. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
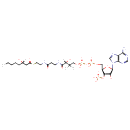
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (S)-3-Hydroxyoctanoyl-CoA | HMDB | | (S)-3-Hydroxyoctanoyl-coenzyme A | HMDB |
|
|---|
| Chemical Formula | C29H50N7O18P3S |
|---|
| Average Molecular Mass | 909.730 g/mol |
|---|
| Monoisotopic Mass | 909.215 g/mol |
|---|
| CAS Registry Number | 79171-48-5 |
|---|
| IUPAC Name | {[(2R,3R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-2-({[hydroxy({hydroxy[(3R)-3-hydroxy-3-({2-[(2-{[(3S)-3-hydroxyoctanoyl]sulfanyl}ethyl)carbamoyl]ethyl}carbamoyl)-2,2-dimethylpropoxy]phosphoryl}oxy)phosphoryl]oxy}methyl)oxolan-3-yl]oxy}phosphonic acid |
|---|
| Traditional Name | [(2R,3R,5R)-5-(6-aminopurin-9-yl)-4-hydroxy-2-({[hydroxy([hydroxy((3R)-3-hydroxy-3-({2-[(2-{[(3S)-3-hydroxyoctanoyl]sulfanyl}ethyl)carbamoyl]ethyl}carbamoyl)-2,2-dimethylpropoxy)phosphoryl]oxy)phosphoryl]oxy}methyl)oxolan-3-yl]oxyphosphonic acid |
|---|
| SMILES | [H][C@](O)(CCCCC)CC(=O)SCCNC(=O)CCNC(=O)[C@]([H])(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H](C(O)[C@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N |
|---|
| InChI Identifier | InChI=1S/C29H50N7O18P3S/c1-4-5-6-7-17(37)12-20(39)58-11-10-31-19(38)8-9-32-27(42)24(41)29(2,3)14-51-57(48,49)54-56(46,47)50-13-18-23(53-55(43,44)45)22(40)28(52-18)36-16-35-21-25(30)33-15-34-26(21)36/h15-18,22-24,28,37,40-41H,4-14H2,1-3H3,(H,31,38)(H,32,42)(H,46,47)(H,48,49)(H2,30,33,34)(H2,43,44,45)/t17-,18+,22?,23-,24-,28+/m0/s1 |
|---|
| InChI Key | ATVGTMKWKDUCMS-FZQVHTIWSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 3-hydroxyacyl coas. These are organic compounds containing a 3-hydroxyl acylated coenzyme A derivative. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
| Direct Parent | 3-hydroxyacyl CoAs |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Pentose phosphate
- Pentose-5-phosphate
- Ribonucleoside 3'-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Monosaccharide phosphate
- Organic pyrophosphate
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Aminopyrimidine
- N-acyl-amine
- N-substituted imidazole
- Organic phosphoric acid derivative
- Monosaccharide
- Fatty amide
- Phosphoric acid ester
- Imidolactam
- Alkyl phosphate
- Pyrimidine
- Imidazole
- Azole
- Heteroaromatic compound
- Tetrahydrofuran
- Carbothioic s-ester
- Thiocarboxylic acid ester
- Amino acid or derivatives
- Carboxamide group
- Secondary alcohol
- Secondary carboxylic acid amide
- Thiocarboxylic acid or derivatives
- Sulfenyl compound
- Organoheterocyclic compound
- Azacycle
- Carboxylic acid derivative
- Oxacycle
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organopnictogen compound
- Carbonyl group
- Primary amine
- Organic oxide
- Alcohol
- Amine
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0891000220-f55700e449c5c748db21 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-1980100000-f8305f14733ea8d55aea | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-2950000000-b772432449eb84aba081 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-05qc-3910031322-a57359169719364b01f7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-2910110010-25e61f1cecb85a279fef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-056r-7900100000-43d6823985b53794e076 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01ox-0100000296-c913e12c204d6a769950 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01p9-3500000290-dcec253505bdee0680b4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-0111900000-12062a5dd2e49f34f086 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0000000029-71ca8cf8e503016684ec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-05vo-9400202683-389c18e28db6571902dd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-056u-4302601921-88fa06ff44f41b042889 | Spectrum | | MS | Mass Spectrum (Electron Ionization) | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0003940 |
|---|
| FooDB ID | FDB023259 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | 28632 |
|---|
| PubChem Compound ID | 53477508 |
|---|
| Kegg Compound ID | C05266 |
|---|
| YMDB ID | YMDB16167 |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|