| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 05:43:30 UTC |
|---|
| Update Date | 2016-11-09 01:21:18 UTC |
|---|
| Accession Number | CHEM035391 |
|---|
| Identification |
|---|
| Common Name | Primapterin |
|---|
| Class | Small Molecule |
|---|
| Description | A member of the class of biopterins that consists of pterin bearing amino, oxo and 1,2-dihydroxypropyl substituents at positions 2, 4 and 7 respectively. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
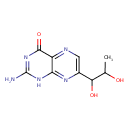
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 7-Biopterin | ChEBI | | 7-Isobiopterin | ChEBI | | L-erythro-7-Isobiopterin | HMDB | | Primapterin, (L-erythro)-isomer | MeSH, HMDB |
|
|---|
| Chemical Formula | C9H11N5O3 |
|---|
| Average Molecular Mass | 237.215 g/mol |
|---|
| Monoisotopic Mass | 237.086 g/mol |
|---|
| CAS Registry Number | 2582-88-9 |
|---|
| IUPAC Name | 2-amino-7-(1,2-dihydroxypropyl)-1,4-dihydropteridin-4-one |
|---|
| Traditional Name | 2-amino-7-(1,2-dihydroxypropyl)-1H-pteridin-4-one |
|---|
| SMILES | CC(O)C(O)C1=CN=C2C(=O)N=C(N)NC2=N1 |
|---|
| InChI Identifier | InChI=1S/C9H11N5O3/c1-3(15)6(16)4-2-11-5-7(12-4)13-9(10)14-8(5)17/h2-3,6,15-16H,1H3,(H3,10,12,13,14,17) |
|---|
| InChI Key | LNXRKRFGNHXANN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pterins and derivatives. These are polycyclic aromatic compounds containing a pterin moiety, which consist of a pteridine ring bearing a ketone and an amine group to form 2-aminopteridin-4(3H)-one. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pteridines and derivatives |
|---|
| Sub Class | Pterins and derivatives |
|---|
| Direct Parent | Pterins and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pterin
- Aminopyrimidine
- Pyrimidone
- Pyrazine
- Pyrimidine
- Vinylogous amide
- Heteroaromatic compound
- 1,2-diol
- Secondary alcohol
- Azacycle
- Aromatic alcohol
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Primary amine
- Organooxygen compound
- Organonitrogen compound
- Organic nitrogen compound
- Amine
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0006-4910000000-b4d6c57ce4057a74cfc1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-0300-7595000000-b436eeb161ee92d9e710 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0079-0090000000-925fbbb5c35d10511d2e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-022i-0590000000-6aec38d83376538e0edd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03di-1900000000-7e663d651c780452a251 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0390000000-b9ad61544ec5c7a678dc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0930000000-0be3f7f3249bf33c601d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9600000000-dd5a13bd6c2574e60f49 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0090000000-0e7e551ea2aecdb52b6f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00di-0190000000-59ecdddec201f0634ae8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-2900000000-c3047f767a1120dd8362 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0930000000-f6d476547533447f8b32 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0900000000-f1b592f0e462c94d10f1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-7900000000-c148cfb5f0996369b85a | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0002263 |
|---|
| FooDB ID | FDB022933 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | 6581 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4590031 |
|---|
| ChEBI ID | 74689 |
|---|
| PubChem Compound ID | 5488923 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. https://www.ncbi.nlm.nih.gov/pubmed/?term=13981581 | | 2. https://www.ncbi.nlm.nih.gov/pubmed/?term=3382399 | | 3. Curtius HC, Adler C, Heizmann C, Blau N, Rebrin I, Ghisla S: 7-substituted pterins: formation and occurrence. J Nutr Sci Vitaminol (Tokyo). 1992;Spec No:501-4. | | 4. Curtius HC, Kuster T, Matasovic A, Blau N, Dhondt JL: Primapterin, anapterin, and 6-oxo-primapterin, three new 7-substituted pterins identified in a patient with hyperphenylalaninemia. Biochem Biophys Res Commun. 1988 Jun 16;153(2):715-21. | | 5. Schallreuter KU, Moore J, Wood JM, Beazley WD, Peters EM, Marles LK, Behrens-Williams SC, Dummer R, Blau N, Thony B: Epidermal H(2)O(2) accumulation alters tetrahydrobiopterin (6BH4) recycling in vitiligo: identification of a general mechanism in regulation of all 6BH4-dependent processes? J Invest Dermatol. 2001 Jan;116(1):167-74. | | 6. Blau N, Kierat L, Matasovic A, Leimbacher W, Heizmann CW, Guardamagna O, Ponzone A: Antenatal diagnosis of tetrahydrobiopterin deficiency by quantification of pterins in amniotic fluid and enzyme activity in fetal and extrafetal tissue. Clin Chim Acta. 1994 May;226(2):159-69. |
|
|---|