| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 05:31:18 UTC |
|---|
| Update Date | 2016-11-09 01:21:15 UTC |
|---|
| Accession Number | CHEM035140 |
|---|
| Identification |
|---|
| Common Name | 5,6-Dihydroxyindole-2-carboxylic acid |
|---|
| Class | Small Molecule |
|---|
| Description | 5,6-Dihydroxyindole-2-carboxylic acid, also known as 5,6-dihydroxyindole-2-carboxylic acid or 5,6-dihydroxyindole-2-carboxylic acid, belongs to the class of organic compounds known as indolecarboxylic acids and derivatives. Indolecarboxylic acids and derivatives are compounds containing a carboxylic acid group (or a derivative thereof) linked to an indole. 5,6-Dihydroxyindole-2-carboxylic acid is possibly soluble (in water) and an extremely weak basic (essentially neutral) compound (based on its pKa). 5,6-Dihydroxyindole-2-carboxylic acid participates in a number of enzymatic reactions, within cattle. In particular, 5,6-Dihydroxyindole-2-carboxylic acid can be biosynthesized from L-dopachrome; which is mediated by the enzyme L-dopachrome tautomerase. In addition, 5,6-Dihydroxyindole-2-carboxylic acid can be converted into melanin through its interaction with the enzyme tyrosinase. In cattle, 5,6-dihydroxyindole-2-carboxylic acid is involved in the metabolic pathway called the tyrosine metabolism pathway. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
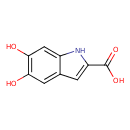
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5,6-DHICA | ChEBI | | 5,6-Dihydroxy-2-indolecarboxylic acid | ChEBI | | 5,6-Dihydroxy-2-indolylcarboxylic acid | ChEBI | | 5,6-Dihydroxyindole-2-carboxylate | ChEBI | | DHI2c | ChEBI | | DHICA | ChEBI | | 5,6-Dihydroxy-2-indolecarboxylate | Generator | | 5,6-Dihydroxy-2-indolylcarboxylate | Generator | | 5,6-Dihydroxy-1H-indole-2-carboxylate | HMDB | | 5,6-Dihydroxy-1H-indole-2-carboxylic acid | HMDB | | 2-Carboxy-5,6-dihydroxyindole | HMDB | | 5,6-Dihydroxy-2-carboxyindole | HMDB | | 5,6-Dihydroxyindole-2-carboxylic acid | HMDB |
|
|---|
| Chemical Formula | C9H7NO4 |
|---|
| Average Molecular Mass | 193.156 g/mol |
|---|
| Monoisotopic Mass | 193.038 g/mol |
|---|
| CAS Registry Number | 4790-08-3 |
|---|
| IUPAC Name | 5,6-dihydroxy-1H-indole-2-carboxylic acid |
|---|
| Traditional Name | dhica |
|---|
| SMILES | OC(=O)C1=CC2=C(N1)C=C(O)C(O)=C2 |
|---|
| InChI Identifier | InChI=1S/C9H7NO4/c11-7-2-4-1-6(9(13)14)10-5(4)3-8(7)12/h1-3,10-12H,(H,13,14) |
|---|
| InChI Key | YFTGOBNOJKXZJC-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as indolecarboxylic acids and derivatives. Indolecarboxylic acids and derivatives are compounds containing a carboxylic acid group (or a derivative thereof) linked to an indole. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Indoles and derivatives |
|---|
| Sub Class | Indolecarboxylic acids and derivatives |
|---|
| Direct Parent | Indolecarboxylic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Indolecarboxylic acid derivative
- Hydroxyindole
- Indole
- Pyrrole-2-carboxylic acid
- Pyrrole-2-carboxylic acid or derivatives
- 1-hydroxy-2-unsubstituted benzenoid
- Substituted pyrrole
- Benzenoid
- Pyrrole
- Heteroaromatic compound
- Azacycle
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00dj-0900000000-86e3bb7f8c4c5358f1ef | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-00du-5329000000-6168733ed8a73f569928 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-002f-0900000000-4323f8e82f9ef2c149e8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0900000000-5180e19b4ff79f8ccbe8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01bc-3900000000-523d85a810268dbe9cf7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0900000000-6d7269d6d89c3eae16f9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0005-0900000000-4d67dcdd279043d44620 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-006t-1900000000-968f54cfbf806d2e2389 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0007-0900000000-7937a0f008a025981516 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0900000000-b0b74fab2c882d47700d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00xs-2900000000-a0ec9715df089ba80b98 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0900000000-b8d69868735567e6ffd7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004l-0900000000-b28b84208bb53ec6a35b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00fu-5900000000-f33eeedd7677021f68df | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0001253 |
|---|
| FooDB ID | FDB022514 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | 1444022 |
|---|
| BioCyc ID | 56-DIHYDROXYINDOLE-2-CARBOXYLATE |
|---|
| METLIN ID | 6110 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 106648 |
|---|
| ChEBI ID | 2003 |
|---|
| PubChem Compound ID | 119405 |
|---|
| Kegg Compound ID | C04185 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Chioccara, Francesco; Novellino, Ettore. Biomimetic synthesis of 5,6-dihydroxyindole-2-carboxylic acid and of its benzyl ester. Synthetic Communications (1987), 17(15), 1815-21. | | 2. Hansson C: Some indolic compounds as markers of the melanocyte activity. Acta Derm Venereol Suppl (Stockh). 1988;138:1-60. | | 3. Yamada K, Walsh N, Hara H, Jimbow K, Chen H, Ito S: Measurement of eumelanin precursor metabolites in the urine as a new marker for melanoma metastases. Arch Dermatol. 1992 Apr;128(4):491-4. | | 4. https://www.ncbi.nlm.nih.gov/pubmed/?term=25450182 |
|
|---|