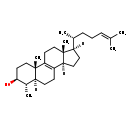
Identification Common Name 4a-Methylzymosterol Class Small Molecule Description A 3beta-sterol that is zymosterol substituted by a 4alpha-methyl group. Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source (3beta,4alpha,5alpha)-4-Methylcholesta-8,24-dien-3-ol ChEBI 4alpha-Methyl-5alpha-cholesta-8,24-dien-3beta-ol ChEBI (3b,4a,5a)-4-Methylcholesta-8,24-dien-3-ol Generator (3β,4α,5α)-4-Methylcholesta-8,24-dien-3-ol Generator 4a-Methyl-5a-cholesta-8,24-dien-3b-ol Generator 4α-Methyl-5α-cholesta-8,24-dien-3β-ol Generator (4S,5S)-4,10,13-Trimethyl-17-(6-methylhept-5-en-2-yl)-2,3,4,5,6,7,11,12,14, 15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol HMDB 4alpha-Methylzymosterol HMDB 4alpha-Methyl-5alpha-cholesta-8(9),24-dien-3beta-ol HMDB 4alpha-Methyl-delta8,24-cholestenol HMDB 4α-Methyl-5α-cholesta-8(9),24-dien-3β-ol HMDB 4α-Methyl-Δ8,24-cholestenol HMDB 4α-Methylzymosterol HMDB
Chemical Formula C28 H46 O Average Molecular Mass 398.664 g/mol Monoisotopic Mass 398.355 g/mol CAS Registry Number 7448-03-5 IUPAC Name (2S,5S,6S,7S,11R,14R,15R)-2,6,15-trimethyl-14-[(2R)-6-methylhept-5-en-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-1(10)-en-5-ol Traditional Name (2S,5S,6S,7S,11R,14R,15R)-2,6,15-trimethyl-14-[(2R)-6-methylhept-5-en-2-yl]tetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-1(10)-en-5-ol SMILES [H][C@@]1(CC[C@@]2([H])C3=C(CC[C@]12C)[C@@]1(C)CC[C@H](O)[C@@H](C)[C@]1([H])CC3)[C@H](C)CCC=C(C)C InChI Identifier InChI=1S/C28H46O/c1-18(2)8-7-9-19(3)22-12-13-24-21-10-11-23-20(4)26(29)15-17-28(23,6)25(21)14-16-27(22,24)5/h8,19-20,22-24,26,29H,7,9-17H2,1-6H3/t19-,20+,22-,23+,24+,26+,27-,28+/m1/s1 InChI Key FOUJWBXBKVVHCJ-YIJYGBTNSA-N