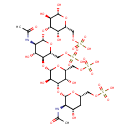
Identification Common Name Keratan Class Small Molecule Description Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source Keratan 6'-sulfate HMDB Keratan 6'-sulphate HMDB Keratan sulfate HMDB Keratan sulphate HMDB Kerato 6'-sulfate HMDB Kerato 6'-sulphate HMDB Kerato sulfate HMDB Kerato sulphate HMDB N-[(2S,3R,4S,6S)-2-{[(2S,3R,4S,5S,6R)-3,5-dihydroxy-2-{[(2R,3S,4R,5R,6S)-4-hydroxy-5-[(1-hydroxyethylidene)amino]-2-[(sulfooxy)methyl]-6-{[(2R,3R,4S,5S,6R)-2,3,5-trihydroxy-6-[(sulfooxy)methyl]oxan-4-yl]oxy}oxan-3-yl]oxy}-6-[(sulfooxy)methyl]oxan-4-yl]oxy}-4-hydroxy-6-[(sulfooxy)methyl]oxan-3-yl]ethanimidate HMDB N-[(2S,3R,4S,6S)-2-{[(2S,3R,4S,5S,6R)-3,5-dihydroxy-2-{[(2R,3S,4R,5R,6S)-4-hydroxy-5-[(1-hydroxyethylidene)amino]-2-[(sulphooxy)methyl]-6-{[(2R,3R,4S,5S,6R)-2,3,5-trihydroxy-6-[(sulphooxy)methyl]oxan-4-yl]oxy}oxan-3-yl]oxy}-6-[(sulphooxy)methyl]oxan-4-yl]oxy}-4-hydroxy-6-[(sulphooxy)methyl]oxan-3-yl]ethanimidate HMDB N-[(2S,3R,4S,6S)-2-{[(2S,3R,4S,5S,6R)-3,5-dihydroxy-2-{[(2R,3S,4R,5R,6S)-4-hydroxy-5-[(1-hydroxyethylidene)amino]-2-[(sulphooxy)methyl]-6-{[(2R,3R,4S,5S,6R)-2,3,5-trihydroxy-6-[(sulphooxy)methyl]oxan-4-yl]oxy}oxan-3-yl]oxy}-6-[(sulphooxy)methyl]oxan-4-yl]oxy}-4-hydroxy-6-[(sulphooxy)methyl]oxan-3-yl]ethanimidic acid HMDB Keratosulfate HMDB Sulfate, keratan HMDB
Chemical Formula C28 H48 N2 O32 S4 Average Molecular Mass 1052.935 g/mol Monoisotopic Mass 1052.107 g/mol CAS Registry Number 69992-87-6 IUPAC Name {[(2S,4S,5R,6S)-5-acetamido-6-{[(2S,3R,4S,5S,6R)-2-{[(2R,3S,4R,5R,6S)-5-acetamido-4-hydroxy-2-[(sulfooxy)methyl]-6-{[(2R,3R,4S,5S,6R)-2,3,5-trihydroxy-6-[(sulfooxy)methyl]oxan-4-yl]oxy}oxan-3-yl]oxy}-3,5-dihydroxy-6-[(sulfooxy)methyl]oxan-4-yl]oxy}-4-hydroxyoxan-2-yl]methoxy}sulfonic acid Traditional Name [(2S,4S,5R,6S)-5-acetamido-6-{[(2S,3R,4S,5S,6R)-2-{[(2R,3S,4R,5R,6S)-5-acetamido-4-hydroxy-2-[(sulfooxy)methyl]-6-{[(2R,3R,4S,5S,6R)-2,3,5-trihydroxy-6-[(sulfooxy)methyl]oxan-4-yl]oxy}oxan-3-yl]oxy}-3,5-dihydroxy-6-[(sulfooxy)methyl]oxan-4-yl]oxy}-4-hydroxyoxan-2-yl]methoxysulfonic acid SMILES CC(=O)N[C@@H]1[C@@H](O)C[C@@H](COS(O)(=O)=O)O[C@H]1O[C@H]1[C@@H](O)[C@@H](COS(O)(=O)=O)O[C@@H](O[C@H]2[C@H](O)[C@@H](NC(C)=O)[C@H](O[C@@H]3[C@@H](O)[C@H](O)O[C@H](COS(O)(=O)=O)[C@@H]3O)O[C@@H]2COS(O)(=O)=O)[C@@H]1O InChI Identifier InChI=1S/C28H48N2O32S4/c1-8(31)29-15-11(33)3-10(4-52-63(40,41)42)56-26(15)62-24-18(35)13(6-54-65(46,47)48)58-28(21(24)38)60-22-14(7-55-66(49,50)51)59-27(16(19(22)36)30-9(2)32)61-23-17(34)12(5-53-64(43,44)45)57-25(39)20(23)37/h10-28,33-39H,3-7H2,1-2H3,(H,29,31)(H,30,32)(H,40,41,42)(H,43,44,45)(H,46,47,48)(H,49,50,51)/t10-,11-,12+,13+,14+,15+,16+,17-,18-,19+,20+,21+,22+,23-,24-,25+,26-,27-,28-/m0/s1 InChI Key KXCLCNHUUKTANI-RBIYJLQWSA-N