| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 05:28:27 UTC |
|---|
| Update Date | 2016-11-09 01:21:14 UTC |
|---|
| Accession Number | CHEM035079 |
|---|
| Identification |
|---|
| Common Name | Stearoyl-CoA |
|---|
| Class | Small Molecule |
|---|
| Description | Stearoyl-CoA is a long-chain acyl CoA ester that acts as an intermediate metabolite in the biosynthesis of monounsaturated fatty acids; a critical committed step in the reaction is the introduction of the cis-configuration double bond into acyl-CoAs (between carbons 9 and 10). This oxidative reaction is catalyzed by the iron-containing, microsomal enzyme, stearoyl-CoA desaturase (SCD, EC 1.14.19.1). NADH supplies the reducing equivalents for the reaction, the flavoprotein is cytochrome b5-reductase and the electron carrier is the heme protein cytochrome b5. Stearoyl-CoA is converted into oleoyl-CoA and then used as a major substrate for the synthesis of various kinds of lipids including phospholipids, triglycerides, cholesteryl esters and wax esters. Oleic acid is the preferred substrate for acyl-CoA cholesterol acyltransferase (ACAT, EC 2.3.1.26) and diacylglycerol acyltransferase (DGAT, EC 2.3.1.20), the enzymes responsible for cholesteryl esters and triglycerides synthesis, respectively. In addition oleate is the major monounsaturated fatty acid in human adipose tissue and in the phospholipid of the red-blood-cell membrane. In the biosynthesis of sphinganine, stearoyl-CoA proceeds through the acyl-CoA + serine -> 3-keto-sphinganine -> sphinganine pathway, with the key enzyme being acyl-CoA serine acyltransferase (EC 2.3.1.50) to yield C20-(3-ketosphinganine) long-chain base. There is growing recognition that acyl-CoA esters could act as signaling molecules in cellular metabolism. (PMID: 12538075, 10998569, Prostaglandins Leukot Essent Fatty Acids. 2003 Feb;68(2):113-21.) [HMDB]. Stearoyl-CoA is found in many foods, some of which are romaine lettuce, grapefruit/pummelo hybrid, radish, and european cranberry. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
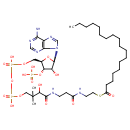
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Octadecanoyl-CoA | HMDB | | Octadecanoyl-coenzyme A | HMDB | | S-Octadecanoate | HMDB | | S-Octadecanoate CoA | HMDB | | S-Octadecanoate coenzyme A | HMDB | | S-Octadecanoic acid | HMDB | | S-Stearate CoA | HMDB | | S-Stearate coenzyme A | HMDB | | S-Stearoylcoenzyme A | HMDB | | Stearoyl coenzyme A | HMDB | | Stearoyl coenzyme A ester | HMDB | | Stearoyl-coenzyme A | HMDB | | Stearyl coenzyme A | HMDB | | Stearyl-CoA | HMDB | | Stearyl-coenzyme A | HMDB |
|
|---|
| Chemical Formula | C39H70N7O17P3S |
|---|
| Average Molecular Mass | 1033.996 g/mol |
|---|
| Monoisotopic Mass | 1033.376 g/mol |
|---|
| CAS Registry Number | 362-66-3 |
|---|
| IUPAC Name | {[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-4-hydroxy-2-({[hydroxy({[hydroxy({3-hydroxy-2,2-dimethyl-3-[(2-{[2-(octadecanoylsulfanyl)ethyl]carbamoyl}ethyl)carbamoyl]propoxy})phosphoryl]oxy})phosphoryl]oxy}methyl)oxolan-3-yl]oxy}phosphonic acid |
|---|
| Traditional Name | [(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-4-hydroxy-2-({[hydroxy([hydroxy(3-hydroxy-2,2-dimethyl-3-[(2-{[2-(octadecanoylsulfanyl)ethyl]carbamoyl}ethyl)carbamoyl]propoxy)phosphoryl]oxy)phosphoryl]oxy}methyl)oxolan-3-yl]oxyphosphonic acid |
|---|
| SMILES | CCCCCCCCCCCCCCCCCC(=O)SCCNC(=O)CCNC(=O)C(O)C(C)(C)COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)(O)=O)N1C=NC2=C1N=CN=C2N |
|---|
| InChI Identifier | InChI=1S/C39H70N7O17P3S/c1-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-30(48)67-23-22-41-29(47)20-21-42-37(51)34(50)39(2,3)25-60-66(57,58)63-65(55,56)59-24-28-33(62-64(52,53)54)32(49)38(61-28)46-27-45-31-35(40)43-26-44-36(31)46/h26-28,32-34,38,49-50H,4-25H2,1-3H3,(H,41,47)(H,42,51)(H,55,56)(H,57,58)(H2,40,43,44)(H2,52,53,54)/t28-,32-,33-,34?,38-/m1/s1 |
|---|
| InChI Key | SIARJEKBADXQJG-KBEKLFCESA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as long-chain fatty acyl coas. These are acyl CoAs where the group acylated to the coenzyme A moiety is a long aliphatic chain of 13 to 21 carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Fatty acyl thioesters |
|---|
| Direct Parent | Long-chain fatty acyl CoAs |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coenzyme a or derivatives
- Purine ribonucleoside 3',5'-bisphosphate
- Purine ribonucleoside bisphosphate
- Purine ribonucleoside diphosphate
- Ribonucleoside 3'-phosphate
- Pentose phosphate
- Pentose-5-phosphate
- Beta amino acid or derivatives
- Glycosyl compound
- N-glycosyl compound
- 6-aminopurine
- Monosaccharide phosphate
- Organic pyrophosphate
- Pentose monosaccharide
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Aminopyrimidine
- Imidolactam
- N-acyl-amine
- N-substituted imidazole
- Organic phosphoric acid derivative
- Monosaccharide
- Pyrimidine
- Alkyl phosphate
- Fatty amide
- Phosphoric acid ester
- Tetrahydrofuran
- Imidazole
- Azole
- Heteroaromatic compound
- Carbothioic s-ester
- Secondary alcohol
- Thiocarboxylic acid ester
- Carboxamide group
- Secondary carboxylic acid amide
- Amino acid or derivatives
- Sulfenyl compound
- Thiocarboxylic acid or derivatives
- Organoheterocyclic compound
- Azacycle
- Oxacycle
- Carboxylic acid derivative
- Organosulfur compound
- Organic oxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic nitrogen compound
- Primary amine
- Organopnictogen compound
- Organic oxide
- Organooxygen compound
- Organonitrogen compound
- Alcohol
- Amine
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-4902010200-7bda8058c8f1f477173c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-1913140000-c5d7727f2a4eb2ce8fdc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000i-1900010000-d8406ea8fe36f8736f7e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00lr-9651431400-a2fe6767c7d15d4d3816 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-5920210000-b2648994ee4ecec2c305 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-057i-5900100000-55fb9ae203fadd900172 | Spectrum | | MS | Mass Spectrum (Electron Ionization) | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0001114 |
|---|
| FooDB ID | FDB022431 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | 1451385 |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | 4242 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Stearoyl-CoA |
|---|
| Chemspider ID | 388366 |
|---|
| ChEBI ID | 15541 |
|---|
| PubChem Compound ID | 439229 |
|---|
| Kegg Compound ID | C00412 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|