Iodate (CHEM035054)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2016-05-26 05:27:05 UTC | ||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:21:14 UTC | ||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM035054 | ||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||
| Common Name | Iodate | ||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||
| Description | Iodate is an iodate is a salt of iodic acid. Because it is more stable than iodide, most health authorities preferentially recommend iodate as an additive to salt for correcting iodine deficiency. Even in a low exposure, doubts have been raised whether the safety of iodate. In humans and rats, oral bioavailability of iodine from iodate is virtually equivalent to that from iodide. When given intravenously to rats, or when added to whole blood or tissue homogenates in vitro or to foodstuff, iodate is quantitatively reduced to iodide by nonenzymatic reactions, and thus becomes available to the body as iodide. Therefore, except perhaps for the gastrointestinal mucosa, exposure of tissues to iodate might be minimal. At much higher doses given intravenously (i.e., above 10 mg/kg), iodate is highly toxic to the retina. Ocular toxicity in humans has occurred only after exposure to doses of 600 to 1,200 mg per individual. Oral exposures of several animal species to high doses, exceeding the human intake from fortified salt by orders of magnitude, pointed to corrosive effects in the gastrointestinal tract, hemolysis, nephrotoxicity, and hepatic injury. Genotoxicity and carcinogenicity data for iodate are scarce or nonexistent. (PMID: 11396703). | ||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type | Not Available | ||||||||||||||||||||||||||||||||||||||||||
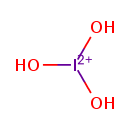
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | IO3 | ||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 174.903 g/mol | ||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 174.889 g/mol | ||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 15454-31-6 | ||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | trihydroxyiodanediium | ||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | trihydroxyiodanediium | ||||||||||||||||||||||||||||||||||||||||||
| SMILES | [O-][I](=O)=O | ||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/HIO3/c2-1(3)4/h(H,2,3,4)/p-1 | ||||||||||||||||||||||||||||||||||||||||||
| InChI Key | ICIWUVCWSCSTAQ-UHFFFAOYSA-M | ||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as other non-metal halides. These are inorganic compounds containing 'other non-metals' and halogen. | ||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | ||||||||||||||||||||||||||||||||||||||||||
| Super Class | Homogeneous non-metal compounds | ||||||||||||||||||||||||||||||||||||||||||
| Class | Other non-metal halides | ||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Other non-metal halides | ||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||
| Origin | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||
| State | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Appearance | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||
| Spectra | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Treatment | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | HMDB0001061 | ||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | FDB022400 | ||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Iodate | ||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | 76615 | ||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 29226 | ||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 84927 | ||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||
| General References | |||||||||||||||||||||||||||||||||||||||||||