| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 05:20:12 UTC |
|---|
| Update Date | 2016-11-09 01:21:13 UTC |
|---|
| Accession Number | CHEM034924 |
|---|
| Identification |
|---|
| Common Name | Dermatan |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
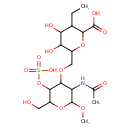
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Ethyl-4,5-dihydroxy-6-[({3-[(1-hydroxyethylidene)amino]-6-(hydroxymethyl)-2-methoxy-5-(sulfooxy)oxan-4-yl}oxy)methyl]oxane-2-carboxylate | Generator | | 3-Ethyl-4,5-dihydroxy-6-[({3-[(1-hydroxyethylidene)amino]-6-(hydroxymethyl)-2-methoxy-5-(sulphooxy)oxan-4-yl}oxy)methyl]oxane-2-carboxylate | Generator | | 3-Ethyl-4,5-dihydroxy-6-[({3-[(1-hydroxyethylidene)amino]-6-(hydroxymethyl)-2-methoxy-5-(sulphooxy)oxan-4-yl}oxy)methyl]oxane-2-carboxylic acid | Generator | | b-Heparin | HMDB | | beta-Heparin | HMDB | | Chondroitin sulfate b | HMDB | | Chondroitin sulfate type b | HMDB | | Chondroitin sulphate b | HMDB | | Chondroitin sulphate type b | HMDB | | Chondroitinsulfuric acid b | HMDB | | Chondroitinsulfuric acid type b | HMDB | | Dermatan 4-sulfate | HMDB | | Dermatan 4-sulphate | HMDB | | Dermatan hydrogen sulfate | HMDB | | Dermatan hydrogen sulphate | HMDB | | Dermatan sulfate | HMDB | | Dermatan sulphate | HMDB | | Desmin 370 | HMDB | | DS 435 | HMDB | | MF 701 | HMDB |
|
|---|
| Chemical Formula | (C16H24NO14S)nC2H6 |
|---|
| Average Molecular Mass | Not Available |
|---|
| Monoisotopic Mass | Not Available |
|---|
| CAS Registry Number | 24967-94-0 |
|---|
| IUPAC Name | 6-({[3-acetamido-6-(hydroxymethyl)-2-methoxy-5-(sulfooxy)oxan-4-yl]oxy}methyl)-3-ethyl-4,5-dihydroxyoxane-2-carboxylic acid |
|---|
| Traditional Name | 6-({[3-acetamido-6-(hydroxymethyl)-2-methoxy-5-(sulfooxy)oxan-4-yl]oxy}methyl)-3-ethyl-4,5-dihydroxyoxane-2-carboxylic acid |
|---|
| SMILES | Not Available |
|---|
| InChI Identifier | InChI=1S/C18H31NO14S/c1-4-8-12(22)13(23)10(31-14(8)17(24)25)6-30-16-11(19-7(2)21)18(29-3)32-9(5-20)15(16)33-34(26,27)28/h8-16,18,20,22-23H,4-6H2,1-3H3,(H,19,21)(H,24,25)(H,26,27,28)/p-1/t8-,9+,10+,11+,12-,13-,14+,15-,16+,18+/m0/s1 |
|---|
| InChI Key | DYTJJIPHSVVNGF-IPUVJUKZSA-M |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as n-acyl-alpha-hexosamines. These are carbohydrate derivatives containing a hexose moiety in which the oxygen atom is replaced by an n-acyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | N-acyl-alpha-hexosamines |
|---|
| Alternative Parents | |
|---|
| Substituents | - N-acyl-alpha-hexosamine
- C-glycosyl compound
- Glycosyl compound
- O-glycosyl compound
- Monosaccharide sulfate
- Monosaccharide
- Oxane
- Pyran
- Sulfuric acid ester
- Alkyl sulfate
- Sulfate-ester
- Sulfuric acid monoester
- Organic sulfuric acid or derivatives
- Secondary alcohol
- Acetal
- Carboximidic acid
- Carboximidic acid derivative
- Carboxylic acid derivative
- Carboxylic acid
- Dialkyl ether
- Ether
- Monocarboxylic acid or derivatives
- Oxacycle
- Propargyl-type 1,3-dipolar organic compound
- Organic 1,3-dipolar compound
- Organoheterocyclic compound
- Organic nitrogen compound
- Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Alcohol
- Organonitrogen compound
- Primary alcohol
- Organopnictogen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0uxr-1200690000-86c93cd9a906d1d39896 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0zn9-1011920000-13d3c57b32ed928f350d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00dl-9210000000-111e8c5a8d3ad36c843e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01b9-1202940000-559e7875e0ef9863af9f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06r2-9547600000-3823d48df58b52c887ff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9200000000-3dd9cd300bcc06b8fee8 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 72682693 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|