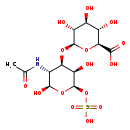
| Chondroitin sulfuric acid | Generator |
| Chondroitin sulphate | Generator |
| Chondroitin sulphuric acid | Generator |
| Chondritinsulfate | HMDB |
| Chondritinsulphate | HMDB |
| Chondroitin 6-sulfate | HMDB |
| Chondroitin 6-sulphate | HMDB |
| Chondroitin polysulfate | HMDB |
| Chondroitin polysulphate | HMDB |
| Chondroitin sodium sulfate ex shark | HMDB |
| Chondroitin sulfate C | HMDB |
| Chondroitin sulfate from swine | HMDB |
| Chondroitin sulphate C | HMDB |
| Chondroitin sulphate from swine | HMDB |
| Chondroitinsulfurate | HMDB |
| Chondroitinsulfuric acid | HMDB |
| Chondroitinsulfuric acids | HMDB |
| Blutal | HMDB |
| Chondroitin 4 sulfate, aluminum salt | HMDB |
| Chondroitin 4-sulfate, aluminum salt | HMDB |
| Chondroitin 6 sulfate, potassium salt | HMDB |
| Chondroitin 6 sulfate, sodium salt | HMDB |
| Chondroitin sulfate a | HMDB |
| Sulfate, sodium chondroitin | HMDB |
| Chondroitin 4-sulfate, potassium salt | HMDB |
| Chondroitin 6-sulfate, potassium salt | HMDB |
| Chondroitin sulfate 4 sulfate, sodium salt | HMDB |
| Chondroitin sulfate, calcium salt | HMDB |
| Chondroitin sulfate, iron (+3) salt | HMDB |
| Chondroitin sulfate, iron salt | HMDB |
| Chondroitin sulfate, potassium salt | HMDB |
| Sulfate, chondroitin | HMDB |
| Translagen | HMDB |
| Chondroitin 4-sulfate | HMDB |
| Chondroitin 6 sulfate | HMDB |
| Chondroitin sulfate, sodium | HMDB |
| Chondroitin sulfate, sodium salt | HMDB |
| Chonsurid | HMDB |
| Sulfates, chondroitin | HMDB |
| Chondroitin 4 sulfate | HMDB |
| Chondroitin 4 sulfate, potassium salt | HMDB |
| Chondroitin 6-sulfate, sodium salt | HMDB |
| Chondroitin sulfate 4-sulfate, sodium salt | HMDB |
| Chondroitin sulfate, zinc salt | HMDB |
| Chondroitin sulfates | HMDB |
| Sodium chondroitin sulfate | HMDB |
| (2S,3S,4S,5R,6R)-6-{[(2R,3R,4R,5R,6R)-2,5-dihydroxy-3-[(1-hydroxyethylidene)amino]-6-(sulfooxy)oxan-4-yl]oxy}-3,4,5-trihydroxyoxane-2-carboxylate | HMDB |
| (2S,3S,4S,5R,6R)-6-{[(2R,3R,4R,5R,6R)-2,5-dihydroxy-3-[(1-hydroxyethylidene)amino]-6-(sulphooxy)oxan-4-yl]oxy}-3,4,5-trihydroxyoxane-2-carboxylate | HMDB |
| (2S,3S,4S,5R,6R)-6-{[(2R,3R,4R,5R,6R)-2,5-dihydroxy-3-[(1-hydroxyethylidene)amino]-6-(sulphooxy)oxan-4-yl]oxy}-3,4,5-trihydroxyoxane-2-carboxylic acid | HMDB |
| Chondroitin sulfate | MeSH |