| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 05:09:15 UTC |
|---|
| Update Date | 2016-11-09 01:21:10 UTC |
|---|
| Accession Number | CHEM034716 |
|---|
| Identification |
|---|
| Common Name | 7-Epijasmonic acid |
|---|
| Class | Small Molecule |
|---|
| Description | An oxylipin that is [(1S)-3-oxocyclopentyl]acetic acid substituted by a (2Z)-pent-2-en-1-yl group at position 2 on the cyclopentanone ring. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
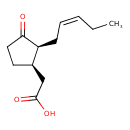
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (+)-Epijasmonic acid | ChEBI | | 2-[(1R,2S)-3-oxo-2-[(Z)-Pent-2-enyl]cyclopentyl]acetic acid | ChEBI | | (+)-Epijasmonate | Generator | | 2-[(1R,2S)-3-oxo-2-[(Z)-Pent-2-enyl]cyclopentyl]acetate | Generator | | (+)-7-Isojasmonate | Generator | | 7-Epijasmonate | Generator |
|
|---|
| Chemical Formula | C12H18O3 |
|---|
| Average Molecular Mass | 210.270 g/mol |
|---|
| Monoisotopic Mass | 210.126 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 2-[(1R,2S)-3-oxo-2-[(2Z)-pent-2-en-1-yl]cyclopentyl]acetic acid |
|---|
| Traditional Name | (+)-7-iso-jasmonic acid |
|---|
| SMILES | CC\C=C/C[C@H]1[C@@H](CC(O)=O)CCC1=O |
|---|
| InChI Identifier | InChI=1S/C12H18O3/c1-2-3-4-5-10-9(8-12(14)15)6-7-11(10)13/h3-4,9-10H,2,5-8H2,1H3,(H,14,15)/b4-3-/t9-,10+/m1/s1 |
|---|
| InChI Key | ZNJFBWYDHIGLCU-QKMQQOOLSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as jasmonic acids. These are lipids containing or derived from a jasmonic acid, with a structure characterized by the presence of an alkene chain linked to a 2-(3-oxocyclopentyl)acetic acid moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Lineolic acids and derivatives |
|---|
| Direct Parent | Jasmonic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Jasmonic acid
- Cyclic ketone
- Ketone
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0920000000-3e2ea863902c1443a94a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0159-9800000000-cae0998084e31d20ae28 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fa6-9100000000-16ffc051b7554d43e070 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0aor-0890000000-0b084a22341872dc4af0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0aou-1940000000-1d0644783ca38085565d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4m-9500000000-8162c16141e7817b5f64 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0gyp-2910000000-a69f966566f2bc59fb69 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001j-5900000000-2ec8ca38efed6833e272 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-005c-9000000000-95226024b29c669c7cfd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9000000000-ec3571c239d0c5c37921 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000000000-396cf428ae175e04137f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9000000000-0ee0c89f8c924a9c618f | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0303804 |
|---|
| FooDB ID | FDB021719 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | CPD-731 |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 5584839 |
|---|
| ChEBI ID | 18435 |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | C16317 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|