| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:51:38 UTC |
|---|
| Update Date | 2016-11-09 01:21:06 UTC |
|---|
| Accession Number | CHEM034360 |
|---|
| Identification |
|---|
| Common Name | 6b-Angeloyl-3b,8b,9b-trihydroxy-7(11)-eremophilen-12,8-olide |
|---|
| Class | Small Molecule |
|---|
| Description | 6b-Angeloyl-3b,8b,9b-trihydroxy-7(11)-eremophilen-12,8-olide is found in green vegetables. 6b-Angeloyl-3b,8b,9b-trihydroxy-7(11)-eremophilen-12,8-olide is a constituent of Petasites japonicus (sweet coltsfoot). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
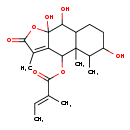
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6,9,9a-Trihydroxy-3,4a,5-trimethyl-2-oxo-2H,4H,4ah,5H,6H,7H,8H,8ah,9H,9ah-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoic acid | HMDB |
|
|---|
| Chemical Formula | C20H28O7 |
|---|
| Average Molecular Mass | 380.432 g/mol |
|---|
| Monoisotopic Mass | 380.184 g/mol |
|---|
| CAS Registry Number | 171422-89-2 |
|---|
| IUPAC Name | 6,9,9a-trihydroxy-3,4a,5-trimethyl-2-oxo-2H,4H,4aH,5H,6H,7H,8H,8aH,9H,9aH-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoate |
|---|
| Traditional Name | 6,9,9a-trihydroxy-3,4a,5-trimethyl-2-oxo-4H,5H,6H,7H,8H,8aH,9H-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoate |
|---|
| SMILES | C\C=C(/C)C(=O)OC1C2=C(C)C(=O)OC2(O)C(O)C2CCC(O)C(C)C12C |
|---|
| InChI Identifier | InChI=1S/C20H28O7/c1-6-9(2)17(23)26-16-14-10(3)18(24)27-20(14,25)15(22)12-7-8-13(21)11(4)19(12,16)5/h6,11-13,15-16,21-22,25H,7-8H2,1-5H3/b9-6+ |
|---|
| InChI Key | MSDFIROCDXOIAM-RMKNXTFCSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as terpene lactones. These are prenol lipids containing a lactone ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene lactones |
|---|
| Direct Parent | Terpene lactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Terpene lactone
- Eremophilanolide or secoeremophilanolide
- Sesquiterpenoid
- Naphthofuran
- Fatty acid ester
- 2-furanone
- Dicarboxylic acid or derivatives
- Fatty acyl
- Cyclic alcohol
- Dihydrofuran
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- Lactone
- Hemiacetal
- Carboxylic acid ester
- Secondary alcohol
- Organoheterocyclic compound
- Oxacycle
- Carboxylic acid derivative
- Polyol
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Organic oxygen compound
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0bwi-9828000000-f5fdbc0a44844a0bcd04 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-001i-9211070000-da192b32324dc665684e | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01q9-2039000000-55917ac26ca1f4c0fd4b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-06si-9143000000-8539669f7d3a6bfe596d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0kur-9300000000-6c181456077dc3e8d218 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-1019000000-cff25006d4898451d956 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-06vs-4029000000-f6ec16d26c1168ecdab9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-054n-7191000000-425b4247b7724ad0f8c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0009000000-43a6402d4255458b3e59 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03e9-0092000000-332fe50fe0bc4d6ee280 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-3921000000-ee057317259ab33e94e9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-9004000000-aeb999bf40dca4c8a4fd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-9002000000-439267d29970209d3e84 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0pb9-9000000000-ea9f932913091753ea73 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0041228 |
|---|
| FooDB ID | FDB021130 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35015133 |
|---|
| ChEBI ID | 175880 |
|---|
| PubChem Compound ID | 131753077 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|