| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:49:09 UTC |
|---|
| Update Date | 2016-11-09 01:21:06 UTC |
|---|
| Accession Number | CHEM034303 |
|---|
| Identification |
|---|
| Common Name | Myrigalone H |
|---|
| Class | Small Molecule |
|---|
| Description | Myrigalone H is found in herbs and spices. Myrigalone H is a constituent of the fruit of Myrica gale (bog myrtle). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,1-Dicyclopropyl-N-(2-oxazolinyl)-methylamine | HMDB | | 1-(2,4-Dihydroxy-6-methoxy-3-methylphenyl)-3-phenyl-1-propanone | HMDB | | 2',4'-Dihydroxy-6'-methoxy-3'-methyldihydrochalcone | HMDB | | 2-((Dicyclopropylmethyl)amino)-2-oxazoline | HMDB | | 2-((Dicyclopropylmethyl)imino)-oxazolidine | HMDB | | 2-((Dicyclopropylmethyl)imino)oxazolidine | HMDB | | 2-(dicyclopropylmethylamino)-2-Oxazoline | HMDB | | 2-(N-(Dicyclopropylmethyl)amino)oxazoline | HMDB | | 2-(N-(Dicyclopropylmethyl)amino)oxazoline phosphate salt | HMDB | | 4,5-dihydro-N-(Dicyclopropylmethyl)-2-oxazolamine | HMDB | | N-(Dicyclopropylmethyl)-4,5-dihydro-1,3-oxazol-2-amine | HMDB | | N-(Dicyclopropylmethyl)-4,5-dihydro-2-oxazolamine | HMDB | | N-(Dicyclopropylmethyl)-4,5-dihydrooxazol-2-amine | HMDB | | Oxaminozoline | HMDB | | Oxaminozoline hemifumarate salt | HMDB | | Rilmenidene | HMDB | | Rilmenidene hemifumarate salt | HMDB | | Rilmenidia | HMDB | | Rilmenidine | HMDB | | Rilmenidine hemifumarate | HMDB | | Rilmenidine hemifumarate salt | HMDB | | Rilmenidinum | HMDB |
|
|---|
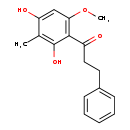
| Chemical Formula | C17H18O4 |
|---|
| Average Molecular Mass | 286.322 g/mol |
|---|
| Monoisotopic Mass | 286.121 g/mol |
|---|
| CAS Registry Number | 143502-00-5 |
|---|
| IUPAC Name | 1-(2,4-dihydroxy-6-methoxy-3-methylphenyl)-3-phenylpropan-1-one |
|---|
| Traditional Name | myrigalone H |
|---|
| SMILES | COC1=C(C(=O)CCC2=CC=CC=C2)C(O)=C(C)C(O)=C1 |
|---|
| InChI Identifier | InChI=1S/C17H18O4/c1-11-14(19)10-15(21-2)16(17(11)20)13(18)9-8-12-6-4-3-5-7-12/h3-7,10,19-20H,8-9H2,1-2H3 |
|---|
| InChI Key | ZDJYWPQCNAPESX-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2'-hydroxy-dihydrochalcones. These are organic compounds containing dihydrochalcone skeleton that carries a hydroxyl group at the 2'-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Linear 1,3-diarylpropanoids |
|---|
| Sub Class | Chalcones and dihydrochalcones |
|---|
| Direct Parent | 2'-Hydroxy-dihydrochalcones |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2'-hydroxy-dihydrochalcone
- Cinnamylphenol
- Alkyl-phenylketone
- Butyrophenone
- Methoxyphenol
- Phenylketone
- Phenoxy compound
- Benzoyl
- O-cresol
- Phenol ether
- Resorcinol
- Aryl ketone
- Aryl alkyl ketone
- Methoxybenzene
- Anisole
- Alkyl aryl ether
- Phenol
- 1-hydroxy-4-unsubstituted benzenoid
- Toluene
- 1-hydroxy-2-unsubstituted benzenoid
- Benzenoid
- Monocyclic benzene moiety
- Vinylogous acid
- Ketone
- Ether
- Organic oxygen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001l-5920000000-7c694791d6166ed8cf65 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-014i-6815900000-8dee2d25494c1072c764 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0290000000-26f432499ef3b8ced191 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-1920000000-55d47809936801ca9023 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-2900000000-38f581d821e8aff414c8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0190000000-fdfcb9e95d023aac3632 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f79-0950000000-eb5bb6ccea2394bd2652 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0674-5910000000-6ecda5de29f39d7ec3df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0290000000-b20320d4a69e2495dd32 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-2960000000-c754614cc99271f96a94 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0043-6940000000-9822c08c31857bf15125 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0090000000-70be716e768197d9205a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0543-2910000000-9b5582b6d710e245c2ff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-4910000000-de862f94160227e3985a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0041169 |
|---|
| FooDB ID | FDB021060 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00008117 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 8260922 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 10085385 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|