| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:44:19 UTC |
|---|
| Update Date | 2016-11-09 01:21:05 UTC |
|---|
| Accession Number | CHEM034198 |
|---|
| Identification |
|---|
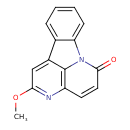
| Common Name | 2-Methoxycanthin-6-one |
|---|
| Class | Small Molecule |
|---|
| Description | 2-Methoxycanthin-6-one is an alkaloid from stem wood of Quassia amara (Surinam quassia). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Methoxy-canthin-6-one | MeSH | | 2-Methoxy-6H-indolo[3,2,1-de][1,5]naphthyridin-6-one, 9ci | HMDB | | 2-Methoxycanthin-6-one | MeSH |
|
|---|
| Chemical Formula | C15H10N2O2 |
|---|
| Average Molecular Mass | 250.252 g/mol |
|---|
| Monoisotopic Mass | 250.074 g/mol |
|---|
| CAS Registry Number | 116353-93-6 |
|---|
| IUPAC Name | 7-methoxy-1,6-diazatetracyclo[7.6.1.0⁵,¹⁶.0¹⁰,¹⁵]hexadeca-3,5(16),6,8,10,12,14-heptaen-2-one |
|---|
| Traditional Name | 7-methoxy-1,6-diazatetracyclo[7.6.1.0⁵,¹⁶.0¹⁰,¹⁵]hexadeca-3,5(16),6,8,10,12,14-heptaen-2-one |
|---|
| SMILES | COC1=NC2=C3N(C4=CC=CC=C4C3=C1)C(=O)C=C2 |
|---|
| InChI Identifier | InChI=1S/C15H10N2O2/c1-19-13-8-10-9-4-2-3-5-12(9)17-14(18)7-6-11(16-13)15(10)17/h2-8H,1H3 |
|---|
| InChI Key | CDWAXMGQLZGHDU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as indolonaphthyridine alkaloids. These are a numerous and relatively straightforward subgroup of the b-carbolines, e.g. Canthin-6-one, in which an additional C3 unit is attached between C-1 and the indole nitrogen to form an additional ring. The group includes a few dimeric examples, such as Haplophytine. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Indolonaphthyridine alkaloids |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Indolonaphthyridine alkaloids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Indolo[3,2-1de][1,5]naphthyridine
- Beta-carboline
- Pyridoindole
- Diazanaphthalene
- Naphthyridine
- Indole
- Indole or derivatives
- Indolizine
- Pyrrolopyridine
- Pyridinone
- Alkyl aryl ether
- Benzenoid
- Pyridine
- Heteroaromatic compound
- Pyrrole
- Lactam
- Ether
- Azacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic oxygen compound
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Organopnictogen compound
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00di-1290000000-fd9a37e6ed952ffd1a02 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0090000000-1ea0d90d2baf66524f9a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0090000000-dda97fbb04fee10f68db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00kf-0920000000-7beb25689d6f03048ebc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0090000000-297c1a3a929bebf819c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0090000000-4dd2d3c074263ffe8565 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00r6-0690000000-d4380b0a64ba4e3cbde3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0090000000-6971a302c8bdd54e5834 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-0090000000-6971a302c8bdd54e5834 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-0090000000-c195d321fa1655d95fe6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0090000000-01844139d9a1e29fbe91 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0090000000-01844139d9a1e29fbe91 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00dj-0790000000-f424cae1c8a12a9a6ea7 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0041064 |
|---|
| FooDB ID | FDB020939 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 2341095 |
|---|
| ChEBI ID | 174291 |
|---|
| PubChem Compound ID | 10106139 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|