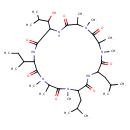
Identification Common Name Ternatin heptapeptide Class Small Molecule Description "Ternatin" is a term used for two unrelated categories of biochemical compounds:
The ternatin heptapeptide — derived from the mushroom Coriolus versicolor
Delphinidin ternatins — derivatives of delphinidin, an anthocyanidin Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source 4',5-Dihydroxy-3,3',7,8-tetramethoxy-flavone MeSH Ternatin (flavonoid) MeSH 4',5-Dihydroxy-3,3',7,8-tetramethoxyflavone MeSH 5,4'-Dihydroxy-3,7,8,3'-tetramethoxyflavone HMDB 5-Hydroxy-2-(4-hydroxy-3-methoxyphenyl)-3,7,8-trimethoxy-4H-1-benzopyran-4-one HMDB cyclo(-OH-Leu-ile-(nme)ala-(nme)leu-leu-(nme)ala-(nme)ala-) HMDB, MeSH Gossypetin 3,7,8,3'-tetramethyl ether HMDB Ternatin a1 HMDB Ternatin heptapeptide HMDB cyclo(-OH-Leucyl-isoleucyl-(N-methyl)alanyl-(N-methyl)leucyl-leucyl-(N-methyl)alanyl-(N-methyl)alanyl-) MeSH, HMDB
Chemical Formula C37 H67 N7 O8 Average Molecular Mass 737.970 g/mol Monoisotopic Mass 737.505 g/mol CAS Registry Number 148619-41-4 IUPAC Name 15-(butan-2-yl)-18-(1-hydroxy-2-methylpropyl)-1,3,4,10,12,13,21-heptamethyl-6,9-bis(2-methylpropyl)-1,4,7,10,13,16,19-heptaazacyclohenicosane-2,5,8,11,14,17,20-heptone Traditional Name 18-(1-hydroxy-2-methylpropyl)-1,3,4,10,12,13,21-heptamethyl-6,9-bis(2-methylpropyl)-15-(sec-butyl)-1,4,7,10,13,16,19-heptaazacyclohenicosane-2,5,8,11,14,17,20-heptone SMILES [H][C@@]1(NC(=O)[C@@H](C)N(C)C(=O)[C@H](C)N(C)C(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C)N(C)C(=O)[C@]([H])(NC1=O)[C@@H](C)CC)[C@H](O)C(C)C InChI Identifier InChI=1S/C37H67N7O8/c1-16-22(8)28-37(52)43(14)25(11)35(50)44(15)27(18-20(4)5)32(47)38-26(17-19(2)3)36(51)42(13)24(10)34(49)41(12)23(9)31(46)40-29(33(48)39-28)30(45)21(6)7/h19-30,45H,16-18H2,1-15H3,(H,38,47)(H,39,48)(H,40,46)/t22-,23+,24-,25-,26-,27-,28+,29+,30+/m0/s1 InChI Key ZMFVAIFXJWEOMH-PTPSPKLBSA-N Spectra Spectra Spectrum Type Description Splash Key View Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive splash10-00di-9000006400-4dce4913f2a9ad8ed9f9 Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_4) - 70eV, Positive Not Available Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-00dr-0000000900-55d2fd913ac031437936 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-00di-1000000900-a0e2db357cb8a93e02ec Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-0pi0-9000000300-c5275daf2469810dc38b Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-01p9-0000007900-c2417cafe903b15bf518 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-08fs-1000009300-d75f40ab7a6b5aec2478 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-06r2-2000009100-e03f67f5ded4ce48b9da Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-000i-0000000900-ec5cb3203a3d54046c14 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-000i-0000003900-0adf5a79207b65569f44 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-074j-3000009000-6170bb2fa3566b43a0a2 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-000i-0000000900-a2cf53b88b12af9c0781 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-000i-0000000900-0f870912f86bcad16e57 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-0nmi-1000009000-99e8ca322f55678f6be4 Spectrum