| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:36:53 UTC |
|---|
| Update Date | 2016-11-09 01:21:03 UTC |
|---|
| Accession Number | CHEM034031 |
|---|
| Identification |
|---|
| Common Name | (all-E)-1,7-bis(4-hydroxyphenyl)-1,4,6-heptatrien-3-one |
|---|
| Class | Small Molecule |
|---|
| Description | (all-E)-1,7-bis(4-hydroxyphenyl)-1,4,6-heptatrien-3-one is found in herbs and spices. (all-E)-1,7-bis(4-hydroxyphenyl)-1,4,6-heptatrien-3-one is a constituent of the rhizomes of Curcuma domestica (turmeric). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
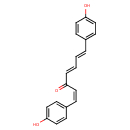
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C19H16O3 |
|---|
| Average Molecular Mass | 292.329 g/mol |
|---|
| Monoisotopic Mass | 292.110 g/mol |
|---|
| CAS Registry Number | 149732-52-5 |
|---|
| IUPAC Name | (1Z,4E,6E)-1,7-bis(4-hydroxyphenyl)hepta-1,4,6-trien-3-one |
|---|
| Traditional Name | (1Z,4E,6E)-1,7-bis(4-hydroxyphenyl)hepta-1,4,6-trien-3-one |
|---|
| SMILES | OC1=CC=C(\C=C\C=C\C(=O)\C=C/C2=CC=C(O)C=C2)C=C1 |
|---|
| InChI Identifier | InChI=1S/C19H16O3/c20-17(10-7-16-8-13-19(22)14-9-16)4-2-1-3-15-5-11-18(21)12-6-15/h1-14,21-22H/b3-1+,4-2+,10-7- |
|---|
| InChI Key | PALMCMYYFAHUGA-ZRFKDBLFSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as linear diarylheptanoids. These are diarylheptanoids with an open heptane chain. The two aromatic rings are linked only by the heptane chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Diarylheptanoids |
|---|
| Sub Class | Linear diarylheptanoids |
|---|
| Direct Parent | Linear diarylheptanoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Linear 1,7-diphenylheptane skeleton
- Hydroxycinnamic acid or derivatives
- Styrene
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Monocyclic benzene moiety
- Benzenoid
- Acryloyl-group
- Enone
- Alpha,beta-unsaturated ketone
- Ketone
- Organic oxide
- Carbonyl group
- Hydrocarbon derivative
- Organooxygen compound
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-006t-0940000000-97f23408da827cb431c1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-00di-6289700000-d8e6898c0bef6261799d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0390000000-4ee3ce306767af8244c3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0564-0920000000-a255d7432339a713b56a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-066r-2920000000-a5cae85958fa21b706db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0190000000-a0af719992f5d44a245e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0690000000-1a9a52afbb065889f355 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-006w-0920000000-1ded92896b73703cd642 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0090000000-2fab2c85c4ca19e57d52 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-0690000000-c80cafd47005c46dc1e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-0950000000-ebd58aad683a96482f1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0090000000-3fe612356dc6bfa0198d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0036-0890000000-c1b768f4d9543d6c5991 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-066r-0950000000-7cbb09047d4067d45438 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0040889 |
|---|
| FooDB ID | FDB020725 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30777515 |
|---|
| ChEBI ID | 172523 |
|---|
| PubChem Compound ID | 131752971 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|