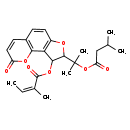| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:28:30 UTC |
|---|
| Update Date | 2016-11-09 01:21:01 UTC |
|---|
| Accession Number | CHEM033842 |
|---|
| Identification |
|---|
| Common Name | 3'-(1''-(3-Methylbutanoyl))-angeloyl vaginidiol |
|---|
| Class | Small Molecule |
|---|
| Description | 3'-(1''-(3-Methylbutanoyl))-angeloyl vaginidiol is found in fats and oils. 3'-(1''-(3-Methylbutanoyl))-angeloyl vaginidiol is a constituent of the roots of Angelica archangelica (angelica). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 8-{2-[(3-methylbutanoyl)oxy]propan-2-yl}-2-oxo-2H,8H,9H-furo[2,3-H]chromen-9-yl (2Z)-2-methylbut-2-enoic acid | HMDB |
|
|---|
| Chemical Formula | C24H28O7 |
|---|
| Average Molecular Mass | 428.475 g/mol |
|---|
| Monoisotopic Mass | 428.184 g/mol |
|---|
| CAS Registry Number | 143061-67-0 |
|---|
| IUPAC Name | 8-{2-[(3-methylbutanoyl)oxy]propan-2-yl}-2-oxo-2H,8H,9H-furo[2,3-h]chromen-9-yl (2Z)-2-methylbut-2-enoate |
|---|
| Traditional Name | 8-{2-[(3-methylbutanoyl)oxy]propan-2-yl}-2-oxo-8H,9H-furo[2,3-h]chromen-9-yl (2Z)-2-methylbut-2-enoate |
|---|
| SMILES | C\C=C(\C)C(=O)OC1C(OC2=C1C1=C(C=C2)C=CC(=O)O1)C(C)(C)OC(=O)CC(C)C |
|---|
| InChI Identifier | InChI=1S/C24H28O7/c1-7-14(4)23(27)30-21-19-16(10-8-15-9-11-17(25)29-20(15)19)28-22(21)24(5,6)31-18(26)12-13(2)3/h7-11,13,21-22H,12H2,1-6H3/b14-7- |
|---|
| InChI Key | HQKAYCHMYMSSEQ-AUWJEWJLSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as angular furanocoumarins. These are furanocoumarins, with a structure characterized by a furan ring angularly fused to a coumarin. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Coumarins and derivatives |
|---|
| Sub Class | Furanocoumarins |
|---|
| Direct Parent | Angular furanocoumarins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Angular furanocoumarin
- Benzopyran
- 1-benzopyran
- Coumaran
- Alkyl aryl ether
- Fatty acid ester
- Pyranone
- Dicarboxylic acid or derivatives
- Pyran
- Fatty acyl
- Benzenoid
- Heteroaromatic compound
- Enoate ester
- Alpha,beta-unsaturated carboxylic ester
- Lactone
- Carboxylic acid ester
- Oxacycle
- Ether
- Carboxylic acid derivative
- Organoheterocyclic compound
- Organic oxide
- Organooxygen compound
- Organic oxygen compound
- Carbonyl group
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4u-9212000000-91224d3a83d1004bc54c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , positive | splash10-004i-0390000000-3cd61da1408a1292cbe0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-9227500000-e69aafe52f0fc28417e8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-055o-9001000000-209bf9fc018d55f369d0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9200000000-59175dd2e76253b66361 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0fc0-7737900000-16323d3835b19cf12778 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0zi3-9536100000-1dd2751ef7cb5ea83c05 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0zgj-9320000000-bf1e69aef0d4c08a0b50 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-4900000000-016bd84a811bb68fd9e6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0kai-9301100000-e7620cb558e6d588e2c0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9100000000-5f2342924015dfafa546 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004j-0095000000-14680a513de181608975 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-2294000000-25a2d00b05439512963e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6v-7191000000-98297096402a6965c1bc | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0040691 |
|---|
| FooDB ID | FDB020493 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 29814600 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 56776263 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|