| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:27:58 UTC |
|---|
| Update Date | 2016-11-09 01:21:00 UTC |
|---|
| Accession Number | CHEM033832 |
|---|
| Identification |
|---|
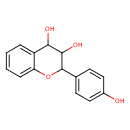
| Common Name | (2S,3R,4R)-3,4,4'-Trihydroxyflavan |
|---|
| Class | Small Molecule |
|---|
| Description | (2S,3R,4R)-3,4,4'-Trihydroxyflavan is found in fruits. (2S,3R,4R)-3,4,4'-Trihydroxyflavan is a constituent of the seeds of Musa sapientum (banana). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4-Methylumbelliferyl-N,n'-diacetyl-beta-chitobiose | HMDB | | 4-Methylumbelliferyl-N,N-diacetylchitobioside | HMDB | | 4-Mudacb | HMDB | | GUM | HMDB | | Me-dinag | HMDB |
|
|---|
| Chemical Formula | C15H14O4 |
|---|
| Average Molecular Mass | 258.269 g/mol |
|---|
| Monoisotopic Mass | 258.089 g/mol |
|---|
| CAS Registry Number | 149747-10-4 |
|---|
| IUPAC Name | 2-(4-hydroxyphenyl)-3,4-dihydro-2H-1-benzopyran-3,4-diol |
|---|
| Traditional Name | 2-(4-hydroxyphenyl)-3,4-dihydro-2H-1-benzopyran-3,4-diol |
|---|
| SMILES | OC1C(O)C2=CC=CC=C2OC1C1=CC=C(O)C=C1 |
|---|
| InChI Identifier | InChI=1S/C15H14O4/c16-10-7-5-9(6-8-10)15-14(18)13(17)11-3-1-2-4-12(11)19-15/h1-8,13-18H |
|---|
| InChI Key | HGNZPAIPVKRUKE-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as leucoanthocyanidins. These are flavonoids consisting of a flavan (3,4-dihydro-2-phenyl-2H-1-benzopyran) moiety that carries two hydroxy groups at the C3- and C4-positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavans |
|---|
| Direct Parent | Leucoanthocyanidins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Leucoanthocyanidin-skeleton
- 3-hydroxyflavonoid
- 4'-hydroxyflavonoid
- 4-hydroxyflavonoid
- Hydroxyflavonoid
- Chromane
- Benzopyran
- 1-benzopyran
- Phenol
- 1-hydroxy-2-unsubstituted benzenoid
- Alkyl aryl ether
- Monocyclic benzene moiety
- Benzenoid
- 1,2-diol
- Secondary alcohol
- Organoheterocyclic compound
- Oxacycle
- Ether
- Organooxygen compound
- Alcohol
- Hydrocarbon derivative
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-0930000000-549a8fc2e62ffda69a30 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-11b9-5425900000-6bed84fb61262afe7605 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0190000000-d10d3bb12067fbddcd77 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ab9-1940000000-ab7a086c698c6a866a88 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6r-9700000000-694e2686ae61bae74d76 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-0190000000-ce7caccb44efb7ce8b01 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ab9-2970000000-b5351f379937670a13ab | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-054o-9600000000-3f6a211cc3dec3331622 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0090000000-7466f0ff4fc176d35adf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-0930000000-ee4dba855d471b8e6e1b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-4900000000-6db2eb115d0f16420f17 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4r-0090000000-e0a309d8eb88e252f016 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-2960000000-0bf8678210a682cb61b6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05mo-4900000000-2e452767f869fcc2f03e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0040679 |
|---|
| FooDB ID | FDB020477 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 32683377 |
|---|
| ChEBI ID | 174379 |
|---|
| PubChem Compound ID | 85979126 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|