| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:21:19 UTC |
|---|
| Update Date | 2016-11-09 01:20:59 UTC |
|---|
| Accession Number | CHEM033689 |
|---|
| Identification |
|---|
| Common Name | Eupafolin 4'-glucoside |
|---|
| Class | Small Molecule |
|---|
| Description | Eupafolin 4'-glucoside is found in herbs and spices. Eupafolin 4'-glucoside is a constituent of rosemary (Rosmarinus officinalis). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
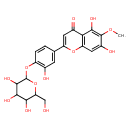
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-[4-(beta-D-Glucopyranosyloxy)-3-hydroxyphenyl]-5,7-dihydroxy-6-methoxy-4H-1-benzopyran-4-one | HMDB | | Nepetin 4'-glucoside | HMDB |
|
|---|
| Chemical Formula | C22H22O12 |
|---|
| Average Molecular Mass | 478.403 g/mol |
|---|
| Monoisotopic Mass | 478.111 g/mol |
|---|
| CAS Registry Number | 112208-83-0 |
|---|
| IUPAC Name | 5,7-dihydroxy-2-(3-hydroxy-4-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-6-methoxy-4H-chromen-4-one |
|---|
| Traditional Name | 5,7-dihydroxy-2-(3-hydroxy-4-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-6-methoxychromen-4-one |
|---|
| SMILES | COC1=C(O)C2=C(OC(=CC2=O)C2=CC(O)=C(OC3OC(CO)C(O)C(O)C3O)C=C2)C=C1O |
|---|
| InChI Identifier | InChI=1S/C22H22O12/c1-31-21-11(26)6-14-16(18(21)28)10(25)5-13(32-14)8-2-3-12(9(24)4-8)33-22-20(30)19(29)17(27)15(7-23)34-22/h2-6,15,17,19-20,22-24,26-30H,7H2,1H3 |
|---|
| InChI Key | FKEFURJFBYTFMP-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as flavonolignans. These are non-conventional lignans that derived from flavonoids. They are characterized by a p-dioxin ring substituted at one carbon atom by a C3C6 (phenylpropan) group and fused to the B-ring of the 2-phenylchromene moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lignans, neolignans and related compounds |
|---|
| Class | Flavonolignans |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Flavonolignans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Flavonolignan
- 3-hydroxyflavone
- 3-hydroxyflavonoid
- 5-hydroxyflavonoid
- 7-hydroxyflavonoid
- Flavone
- Hydroxyflavonoid
- 2-phenylbenzo-1,4-dioxane
- Phenylbenzodioxane
- Chromone
- Benzo-1,4-dioxane
- Benzodioxane
- Benzopyran
- Methoxyphenol
- 1-benzopyran
- Phenoxy compound
- Anisole
- Methoxybenzene
- Phenol ether
- Phenol
- Pyranone
- Alkyl aryl ether
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Para-dioxin
- Pyran
- Benzenoid
- Monocyclic benzene moiety
- Vinylogous acid
- Heteroaromatic compound
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Ether
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Primary alcohol
- Organooxygen compound
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-08mi-9303700000-f70cde1bbaf25301f61a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-0059-2510019000-f1abb21ab940e4bbccee | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-02vi-0038900000-2fef1b2c1ec10629a0ec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0289100000-4b005ae4aeaf471afe3a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0aos-1695000000-7518e354f35b6a5d07a1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00or-1204900000-dfbdc647cd67f986896b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-1379400000-603591d9cc6c75c8ed28 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01bd-2192000000-9fbf8d5f31778e66c6ef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000900000-09b9d71f10bec0f8414f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0000900000-09b9d71f10bec0f8414f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-003r-0941800000-a9a07e86c0b379aedcd6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0000900000-2055daa3382fd02da593 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-004i-0000900000-ed50fe21b91d5e47a3f2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0019-0509300000-079e89f6c060e191b2c6 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0040510 |
|---|
| FooDB ID | FDB020275 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00013693 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 74977888 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|