| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 04:15:48 UTC |
|---|
| Update Date | 2016-11-09 01:20:57 UTC |
|---|
| Accession Number | CHEM033565 |
|---|
| Identification |
|---|
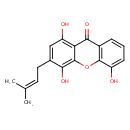
| Common Name | 1,4,5-Trihydroxy-3-prenylxanthone |
|---|
| Class | Small Molecule |
|---|
| Description | 1,4,5-Trihydroxy-3-prenylxanthone is found in fruits. 1,4,5-Trihydroxy-3-prenylxanthone is a constituent of the root bark of Garcinia livingstonei (imbe). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,4,5-Trihydroxy-3-(3-methyl-2-butenyl)-9H-xanthen-9-one, 9ci | HMDB |
|
|---|
| Chemical Formula | C18H16O5 |
|---|
| Average Molecular Mass | 312.317 g/mol |
|---|
| Monoisotopic Mass | 312.100 g/mol |
|---|
| CAS Registry Number | 136364-72-2 |
|---|
| IUPAC Name | 1,4,5-trihydroxy-3-(3-methylbut-2-en-1-yl)-9H-xanthen-9-one |
|---|
| Traditional Name | 1,4,5-trihydroxy-3-(3-methylbut-2-en-1-yl)xanthen-9-one |
|---|
| SMILES | CC(C)=CCC1=CC(O)=C2C(=O)C3=C(OC2=C1O)C(O)=CC=C3 |
|---|
| InChI Identifier | InChI=1S/C18H16O5/c1-9(2)6-7-10-8-13(20)14-16(22)11-4-3-5-12(19)17(11)23-18(14)15(10)21/h3-6,8,19-21H,7H2,1-2H3 |
|---|
| InChI Key | BRVVGOBMRRGKCK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as xanthones. These are polycyclic aromatic compounds containing a xanthene moiety conjugated to a ketone group at carbon 9. Xanthene is a tricyclic compound made up of two benzene rings linearly fused to each other through a pyran ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzopyrans |
|---|
| Sub Class | 1-benzopyrans |
|---|
| Direct Parent | Xanthones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Xanthone
- Prenylbenzoquinol
- Chromone
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Pyranone
- Pyran
- Benzenoid
- Heteroaromatic compound
- Vinylogous acid
- Polyol
- Oxacycle
- Organic oxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001i-2090000000-3f35101f924661aa14df | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-03di-2102690000-82249fc53bcf6ad7e02c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0049000000-0f1756f093e355cc07f1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-5092000000-e532fb236bd1e4ee36e8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052r-9410000000-86c3d602b53bde7dbad9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0009000000-c755f8aff099223bf970 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0029000000-9b8e89d3f948b7c690c3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4r-3930000000-c3f993ef2daf77bbc2b7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0039000000-967c66db00050282103b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ab9-0091000000-5f9561b2f5d0c1aadd08 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-056u-1190000000-81a1ac4e344bf5e2c9cb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0009000000-a968dea78994eb63447d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0049000000-461dcdcaa86ee90fccad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-08i0-1970000000-a0dadeae5cc5a360e974 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0040354 |
|---|
| FooDB ID | FDB020080 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00057863 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 23252000 |
|---|
| ChEBI ID | 174994 |
|---|
| PubChem Compound ID | 15127377 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|