| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:50:12 UTC |
|---|
| Update Date | 2016-11-09 01:20:49 UTC |
|---|
| Accession Number | CHEM032960 |
|---|
| Identification |
|---|
| Common Name | Physalin P |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
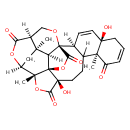
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C28H30O10 |
|---|
| Average Molecular Mass | 526.538 g/mol |
|---|
| Monoisotopic Mass | 526.184 g/mol |
|---|
| CAS Registry Number | 149204-27-3 |
|---|
| IUPAC Name | (1S,2R,5R,9S,14R,17S,18R,21R,24S,26R,27R)-5,14-dihydroxy-2,9,26-trimethyl-3,19,23,28-tetraoxaoctacyclo[16.9.2.0¹,⁵.0²,²⁴.0⁸,¹⁷.0⁹,¹⁴.0¹⁸,²⁷.0²¹,²⁶]nonacosa-11,15-diene-4,10,22,29-tetrone |
|---|
| Traditional Name | (1S,2R,5R,9S,14R,17S,18R,21R,24S,26R,27R)-5,14-dihydroxy-2,9,26-trimethyl-3,19,23,28-tetraoxaoctacyclo[16.9.2.0¹,⁵.0²,²⁴.0⁸,¹⁷.0⁹,¹⁴.0¹⁸,²⁷.0²¹,²⁶]nonacosa-11,15-diene-4,10,22,29-tetrone |
|---|
| SMILES | [H][C@@]12[C@]34OC[C@]5([H])C(=O)O[C@@]([H])(C[C@]15C)[C@@]1(C)OC(=O)[C@@](O)(CCC5([H])[C@]3([H])C=C[C@]3(O)CC=CC(=O)[C@@]53C)[C@@]21OC4=O |
|---|
| InChI Identifier | InChI=1S/C28H30O10/c1-22-11-17-24(3)28-19(22)27(21(32)38-28,35-12-15(22)18(30)36-17)14-6-9-25(33)8-4-5-16(29)23(25,2)13(14)7-10-26(28,34)20(31)37-24/h4-6,9,13-15,17,19,33-34H,7-8,10-12H2,1-3H3/t13?,14-,15+,17-,19-,22-,23+,24+,25+,26-,27+,28+/m0/s1 |
|---|
| InChI Key | LDYGFZHEHXLMTL-GJFGNQQKSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as physalins and derivatives. These are steroidal constituents of Physalis plants which possess an unusual 13,14-seco-16,24-cyclo-steroidal ring skeleton (where the bond that is normally present between the 13 and 14 positions in other steroids is broken while a new bond between positions 16 and 24 is formed). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Physalins and derivatives |
|---|
| Direct Parent | Physalins and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Physalin skeleton
- Tricarboxylic acid or derivatives
- Furopyran
- Cyclohexenone
- Delta_valerolactone
- Delta valerolactone
- Pyran
- Oxane
- Gamma butyrolactone
- Tetrahydrofuran
- Tertiary alcohol
- Furan
- Cyclic alcohol
- Lactone
- Ketone
- Carboxylic acid ester
- Oxacycle
- Organoheterocyclic compound
- Ether
- Dialkyl ether
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a6r-0000390000-6cad9f4aaacee966d392 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a6u-1101890000-fbe06158b1edb396468e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6x-3004900000-4afa9ed0a8369a46cd69 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0000390000-8274f674e1387d26b14f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-057i-0000790000-eed87204c16b6e9c4079 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udr-0140900000-d97ca642a26feee29998 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|