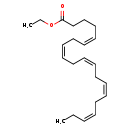
| Synonyms | | Value | Source |
|---|
| (5Z,8Z,11Z,14Z,17Z)-Eicosapentaenoic acid ethyl ester | ChEBI | | (all-Z)-5,8,11,14,17-Eicosapentaenoic acid ethyl ester | ChEBI | | all-cis-Ethyl 5,8,11,14,17-icosapentaenoate | ChEBI | | AMR 101 | ChEBI | | AMR101 | ChEBI | | cis-Eicosapentaenoic acid ethyl ester | ChEBI | | e-EPA | ChEBI | | Eicosapentaenoic acid ethyl ester | ChEBI | | EPA ethyl ester | ChEBI | | Epadel | ChEBI | | Ethyl (5Z,8Z,11Z,14Z,17Z)-eicosa-5,8,11,14,17-pentaenoate | ChEBI | | Ethyl (5Z,8Z,11Z,14Z,17Z)-eicosapentaenoate | ChEBI | | Ethyl (all cis)-5,8,11,14,17-icosapentaenoate | ChEBI | | Ethyl all-cis-5,8,11,14,17-icosapentaenoate | ChEBI | | Ethyl eicosapentaenoate | ChEBI | | Icosapent | ChEBI | | Icosapent ethyl | ChEBI | | Timnodonic acid ethyl ester | ChEBI | | Vascepa | ChEBI | | Epadel S | Kegg | | (5Z,8Z,11Z,14Z,17Z)-Eicosapentaenoate ethyl ester | Generator | | (all-Z)-5,8,11,14,17-Eicosapentaenoate ethyl ester | Generator | | all-cis-Ethyl 5,8,11,14,17-icosapentaenoic acid | Generator | | cis-Eicosapentaenoate ethyl ester | Generator | | Eicosapentaenoate ethyl ester | Generator | | Ethyl (5Z,8Z,11Z,14Z,17Z)-eicosa-5,8,11,14,17-pentaenoic acid | Generator | | Ethyl (5Z,8Z,11Z,14Z,17Z)-eicosapentaenoic acid | Generator | | Ethyl (all cis)-5,8,11,14,17-icosapentaenoic acid | Generator | | Ethyl all-cis-5,8,11,14,17-icosapentaenoic acid | Generator | | Ethyl eicosapentaenoic acid | Generator | | Timnodonate ethyl ester | Generator | | Ethyl icosapentic acid | Generator | | (5Z,8Z,11Z,14Z,17Z)-Eicosapetaenoic acid ethyl ester | ChEBI | | (5Z,8Z,11Z,14Z,17Z)-Eicosapetaenoate ethyl ester | Generator | | Eicosapentaenoic acid, ethyl ester | HMDB | | Epa-e | HMDB, MeSH | | Ethyl epa | HMDB | | Ethyl ester(all-Z)-5,8,11,14,17-eicosapentaenoic acid | HMDB | | Ethyl icosapentaenoate | HMDB, MeSH | | Ethyl icosapentate, jan | HMDB | | MND 21 | HMDB | | AMR-101 | MeSH | | Ethyl (5Z,8Z,11Z,14Z,17Z)-icosa-5,8,11,14,17-pentaenoate | MeSH | | Ethyl icosapentate | ChEBI |
|
|---|
| InChI Identifier | InChI=1S/C22H34O2/c1-3-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19-20-21-22(23)24-4-2/h5-6,8-9,11-12,14-15,17-18H,3-4,7,10,13,16,19-21H2,1-2H3/b6-5-,9-8-,12-11-,15-14-,18-17- |
|---|