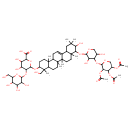| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:42:47 UTC |
|---|
| Update Date | 2016-11-09 01:20:47 UTC |
|---|
| Accession Number | CHEM032807 |
|---|
| Identification |
|---|
| Common Name | Acetylsoyasaponin A5 |
|---|
| Class | Small Molecule |
|---|
| Description | Acetylsoyasaponin A5 is found in pulses. Acetylsoyasaponin A5 is a constituent of soybean (Glycine max). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6-({9-[(3,5-dihydroxy-4-{[3,4,5-tris(acetyloxy)oxan-2-yl]oxy}oxan-2-yl)oxy]-10-hydroxy-4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,14,14a,14b-icosahydropicen-3-yl}oxy)-3,4-dihydroxy-5-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxane-2-carboxylate | HMDB |
|
|---|
| Chemical Formula | C58H90O26 |
|---|
| Average Molecular Mass | 1203.320 g/mol |
|---|
| Monoisotopic Mass | 1202.572 g/mol |
|---|
| CAS Registry Number | 117230-34-9 |
|---|
| IUPAC Name | 6-({9-[(3,5-dihydroxy-4-{[3,4,5-tris(acetyloxy)oxan-2-yl]oxy}oxan-2-yl)oxy]-10-hydroxy-4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,14,14a,14b-icosahydropicen-3-yl}oxy)-3,4-dihydroxy-5-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxane-2-carboxylic acid |
|---|
| Traditional Name | 6-({9-[(3,5-dihydroxy-4-{[3,4,5-tris(acetyloxy)oxan-2-yl]oxy}oxan-2-yl)oxy]-10-hydroxy-4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-1,2,3,4a,5,6,7,8,9,10,12,12a,14,14a-tetradecahydropicen-3-yl}oxy)-3,4-dihydroxy-5-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxane-2-carboxylic acid |
|---|
| SMILES | CC(=O)OC1COC(OC2C(O)COC(OC3C(O)C(C)(C)CC4C5=CCC6C7(C)CCC(OC8OC(C(O)C(O)C8OC8OC(CO)C(O)C(O)C8O)C(O)=O)C(C)(CO)C7CCC6(C)C5(C)CCC34C)C2O)C(OC(C)=O)C1OC(C)=O |
|---|
| InChI Identifier | InChI=1S/C58H90O26/c1-24(61)76-31-22-75-51(45(78-26(3)63)42(31)77-25(2)62)81-41-29(64)21-74-49(40(41)70)84-47-46(71)53(4,5)19-28-27-11-12-33-55(7)15-14-34(56(8,23-60)32(55)13-16-58(33,10)57(27,9)18-17-54(28,47)6)80-52-44(38(68)37(67)43(82-52)48(72)73)83-50-39(69)36(66)35(65)30(20-59)79-50/h11,28-47,49-52,59-60,64-71H,12-23H2,1-10H3,(H,72,73) |
|---|
| InChI Key | ONYGLIKHHLDSEF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as steroidal saponins. These are saponins in which the aglycone moiety is a steroid. The steroidal aglycone is usually a spirostane, furostane, spirosolane, solanidane, or curcubitacin derivative. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Steroidal glycosides |
|---|
| Direct Parent | Steroidal saponins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Steroidal saponin
- Diterpene glycoside
- Furostane-skeleton
- 22-hydroxysteroid
- Diterpenoid
- Hydroxysteroid
- Terpene glycoside
- Delta-5-steroid
- Fatty acyl glycoside
- Fatty acyl glycoside of mono- or disaccharide
- Alkyl glycoside
- Disaccharide
- O-glycosyl compound
- Glycosyl compound
- Fatty acyl
- Oxane
- Tetrahydrofuran
- Secondary alcohol
- Hemiacetal
- Organoheterocyclic compound
- Polyol
- Acetal
- Oxacycle
- Organooxygen compound
- Alcohol
- Organic oxygen compound
- Primary alcohol
- Hydrocarbon derivative
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01rm-3900102365-e08213e904143f09c394 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-06xt-2400508897-1dd8a7a465cb6bcea83a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-06yk-1610509553-226eeb6c4a9a566e1886 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a59-7920000031-1da9ffdcec0bb1bf4bc6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0bvi-6910000132-b13f0adb9df2c135293c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9300001010-e50515e5a69d46ee45ef | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zgi-8972000000-90955803b42b2ddaff89 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4l-6940000315-aa1d39c380a78d3e1d37 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052f-9310000001-5bea4ca0bd2a8885bd4e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-1690000130-d2743b0327e6c0d2cc92 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0f79-0690526400-b80d4777eaf8c579ec0f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ldi-2930021000-7c6f20e674a15482dc4d | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0039480 |
|---|
| FooDB ID | FDB019084 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00054753 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 14186217 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|